Self Assess 2.4:
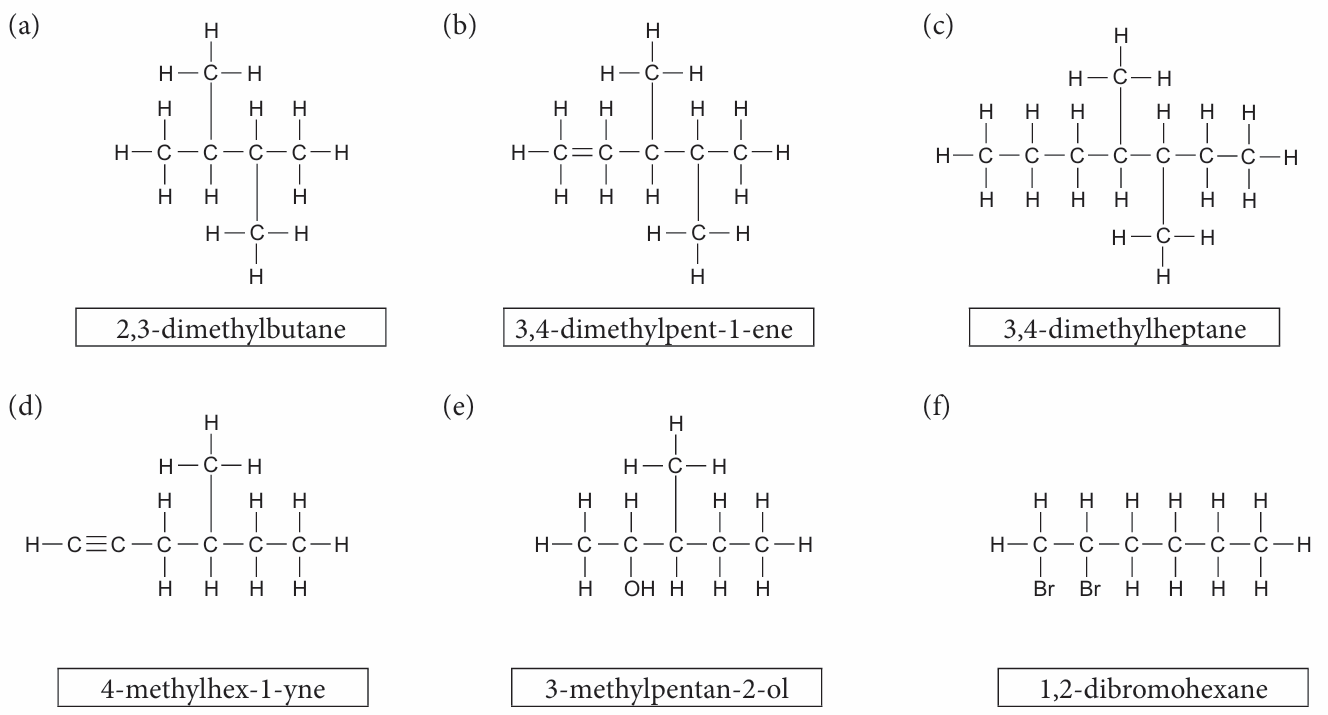
1. Draw the structural formula for each of the following compounds:
(a) 2,3-dimethylbutane
(b) 3,4-dimethylpent-1-ene
(c) 3,4-dimethylheptane
(d) 4-methylhex-1-yne
(e) 3-methylpentan-2-ol
(f) 1,2-dibromohexane
1. Draw the structural formula for each of the following compounds:
(a) 2,3-dimethylbutane
(b) 3,4-dimethylpent-1-ene
(c) 3,4-dimethylheptane
(d) 4-methylhex-1-yne
(e) 3-methylpentan-2-ol
(f) 1,2-dibromohexane
2. Figure 2.24 shows the structural formulae of three hydrocarbons, X, Y and Z.
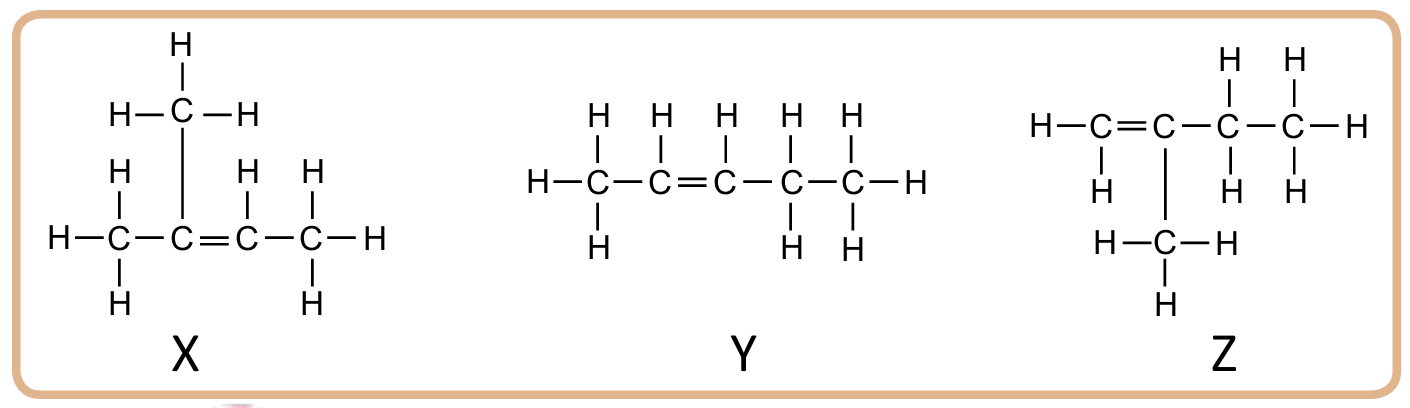
(a) Name the hydrocarbons labeled X, Y and Z.
(b) Determine whether X, Y and Z are isomers. Give your reasons.
Answer:
1.
2. (a)
X : 2-methylbut-2-ene
Y : Pent-2-ene
Z : 2-methylbut-1-ene
(b)
– X and Z are isomers
– X and Z have different structural formulae but the same molecular formula which is C5H10
