The following figure shows classifications of carbon compounds.
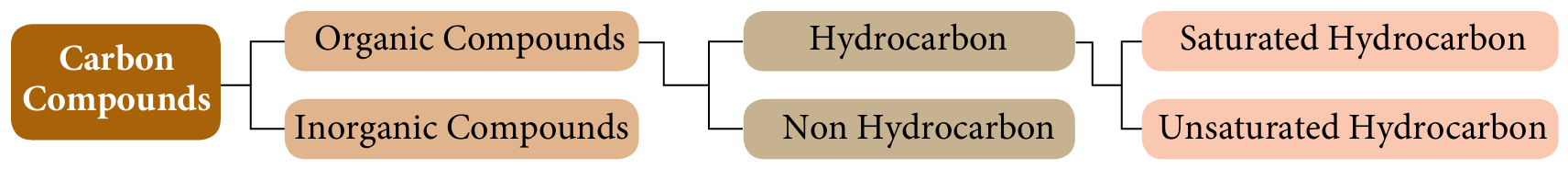
Based on the figure above, state the definitions of:
(a) Organic compounds.
(b) Hydrocarbon and non hydrocarbon.
(c) Saturated and unsaturated hydrocarbons.
Answer:
(a) Carbon compounds are compounds that contain carbon as their constituent element.
(b)
– Organic compounds that contain only hydrogen and carbon.
– Organic compounds that contain carbon and hydrogen and other elements, such as oxygen, nitrogen, phosphorus or halogen.
(c)
– Hydrocarbons that contain only single bonds between carbon atoms.
– Hydrocarbons that contain at least one double or triple bond between carbon atoms.
(a) What is meant by cracking?
(b) Copy and complete the following reactions:
(i) C10H22 → C6H14 + …………..
(ii) ………….. → C4H8 + C3H6 + C4H12
(c) Discuss the importance of the cracking process.
Answer:
(a) Cracking is the process of breaking long chain hydrocarbons into smaller hydrocarbons at a high temperature with the presence of a catalyst.
(b)(i) C10H22 → C6H14 + C4H8
(b)(ii) C11H26 → C4H8 + C3H6 + C4H12
(c)
– The demand for small sized hydrocarbons is higher.
– The separation of petroleum into its fractions by fractional distillation does not meet the demand for small-sized hydrocarbons.
– The cracking process produces smaller sized hydrocarbons that can be used as fuel as well as raw materials in the petrochemical industry.
