Figure 1 shows the products of petroleum fractional distillation at the oil refinery.
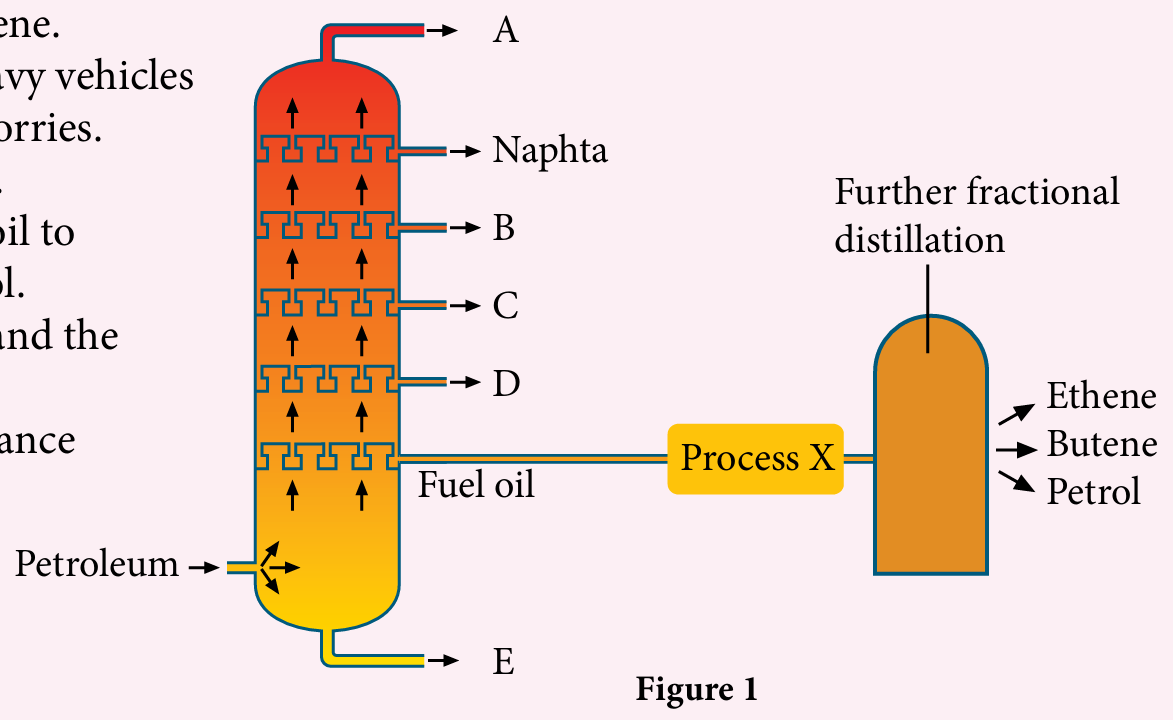
(a) Why can petroleum be separated into its components by fractional distillation ?
(b) Which of the following is A, B, C, D or E?
(i) A fraction of kerosene.
(ii) Used as fuel for heavy vehicles such as buses and lorries.
(iii) Used to pave roads.
(c) Process X converts fuel oil to ethene, butene and petrol.
(i) What is process X and the catalyst used?
(ii) What is the importance of process X?
Answer:
(a)
– Petroleum is a mixture of simple or long chain hydrocarbons.
– Fractions in petroleum can be separated because each hydrocarbon fraction has its own boiling point.
(b)(i) B
(b)(ii) D
(b)(iii) E
(c)(i)
– The X process is the cracking process.
– The catalyst is a mixture of silicon (IV) oxide and aluminum oxide.
(c)(ii)
– The cracking process produces smaller sized hydrocarbons that can be used as fuel as well as raw materials in the petrochemical industry.
Figure 2 shows the structural formula of compound X.
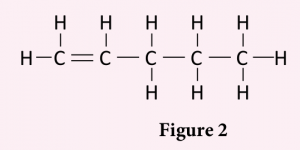
(a) Compound X is an unsaturated hydrocarbon.
(i) What is meant by unsaturated hydrocarbon?
(ii) State the homologous series of compound X.
(iii) Compound X forms an isomer.
Draw the structural formulae of all isomers for compound X and name each isomer according to the IUPAC nomenclature.
(b) At a temperature of 180 °C and with the presence of nickel as a catalyst, compound X can be converted into saturated hydrocarbons.
(i) Name the reaction that takes place.
(ii) Draw the structural formula for the compound formed
(c) Compound X burns completely in excess oxygen.
(i) Write a balanced chemical equation for a complete combustion of compound X.
(ii) 14 g of compound X undergoes complete combustion at room temperature.
Calculate the volume of carbon dioxide gas released.
[1 mole of gas occupies a volume of 24 dm3 at room temperature. Relative atomic mass: H = 1; C = 12].
Answer:
(a)(i) – Hydrocarbons that have at least one double or triple bond between carbon atoms.
(a)(ii) Alkene
(a)(iii)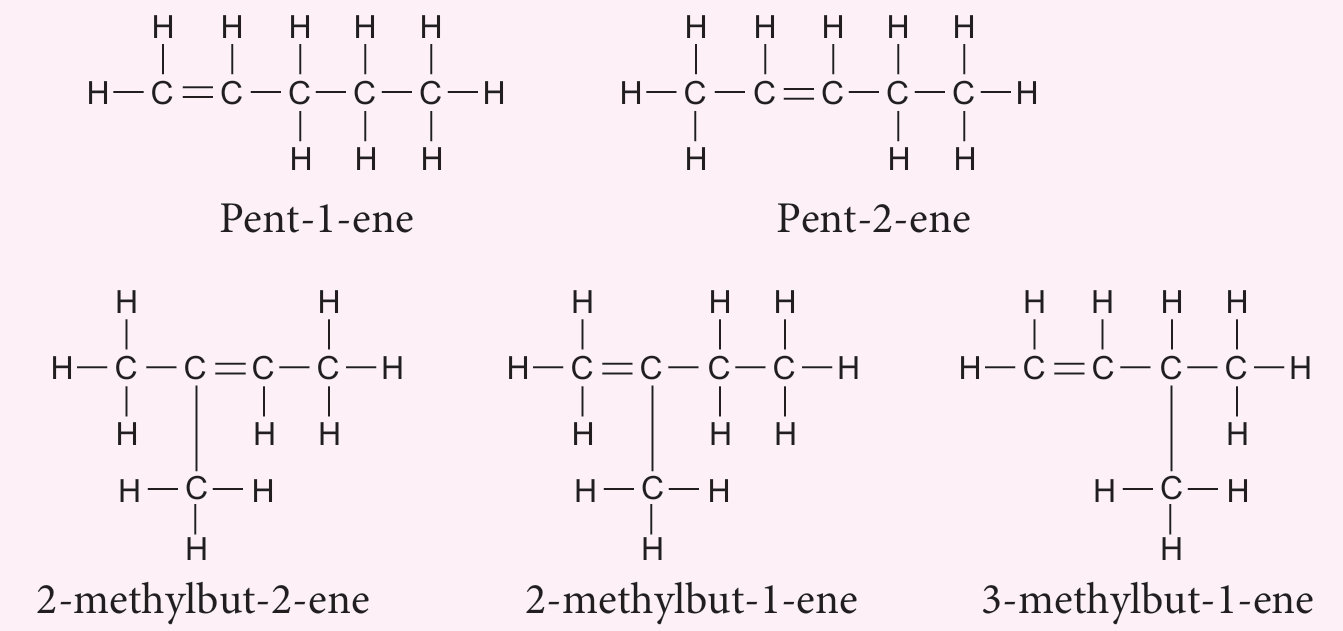
(b)(i) Addition of hydrogen (Hydrogenation)
(b)(ii)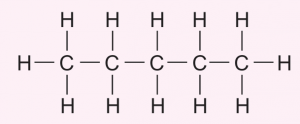
(c)(i) $$
2 \mathrm{C}_5 \mathrm{H}_{10}+15 \mathrm{O}_2 \rightarrow 10 \mathrm{CO}_2+10 \mathrm{H}_2 \mathrm{O}
$$
(c)(ii) $$
\begin{aligned}
\text { Number of } \mathrm{mol} \mathrm{X} & =\frac{14 \mathrm{~g}}{70 \mathrm{~g} \mathrm{~mol}^{-1}} \\
& =0.2 \mathrm{~mol}
\end{aligned}
$$
From the equation: 2 mol C5H10 : 10 mol CO2
0.2 mol C5H10 : 1.0 mol CO2
$$
\begin{aligned}
\text { Volume of carbon dioxide } & =1.0 \mathrm{~mol} \times 24 \mathrm{dm}^3 \mathrm{~mol}^{-1} \\
& =24 \mathrm{dm}^3
\end{aligned}
$$
Figure 3 shows a series of reactions involving compound Q, C2H6O.
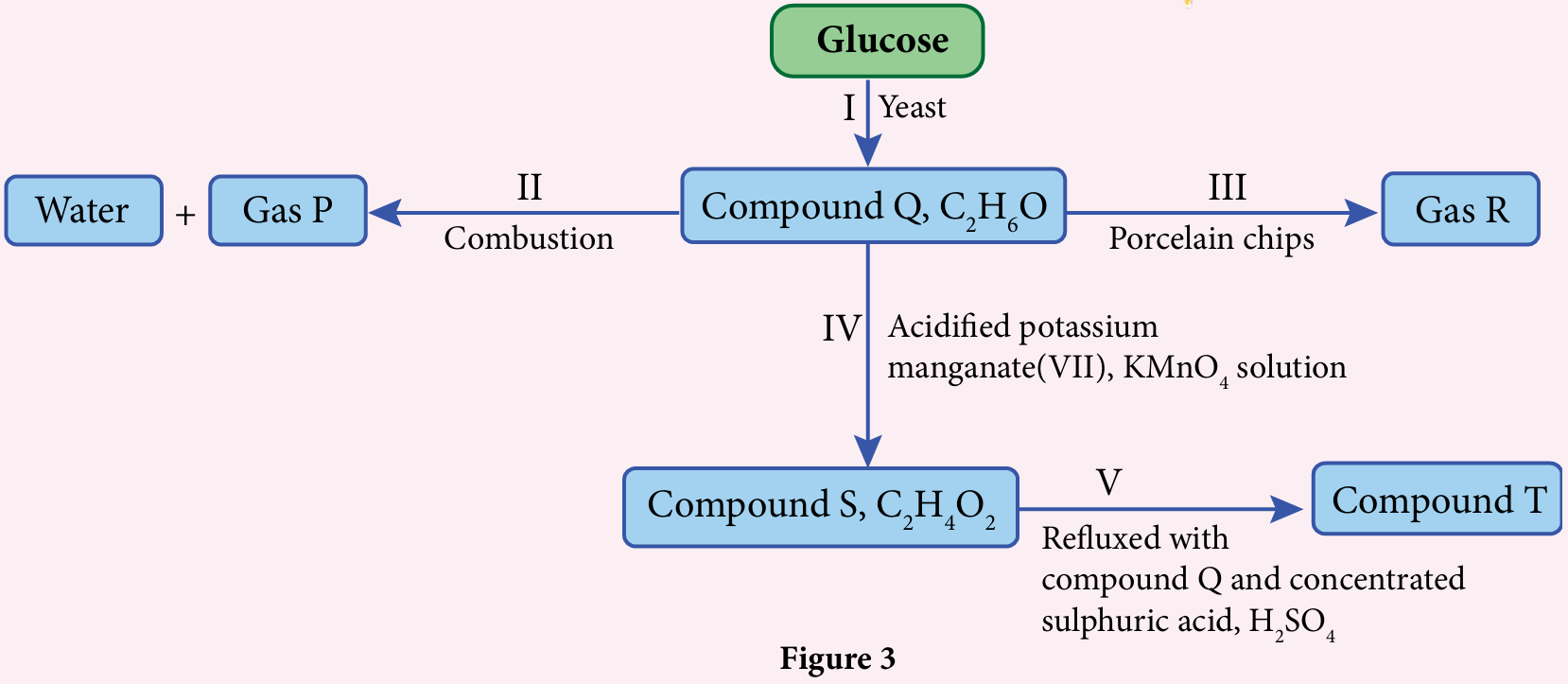
(a) (i) Name reaction I.
(ii) State the name of compound Q.
(iii) Draw the structural formula of compound Q.
(b) Write the chemical equation for the complete combustion of compound Q.
(c) (i) State the name of gas R released in reaction III.
(ii) Draw the apparatus set-up to carry out the experiment for reaction III in the laboratory.
(d) State the colour change of acidified potassium manganate(VII), KMnO4 solution in reaction IV.
(e) (i) State the name of reaction V.
(ii) State the name of compound T.
Answer:
(a)(i) Fermentation
(a)(ii) Ethanol
(a)(iii)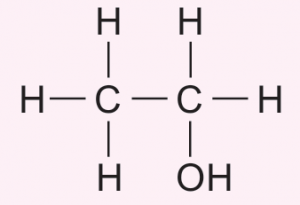
(b) C2H5OH + 3O2 → 2CO2+ 3H2O
(c)(i) Ethene
(c)(ii)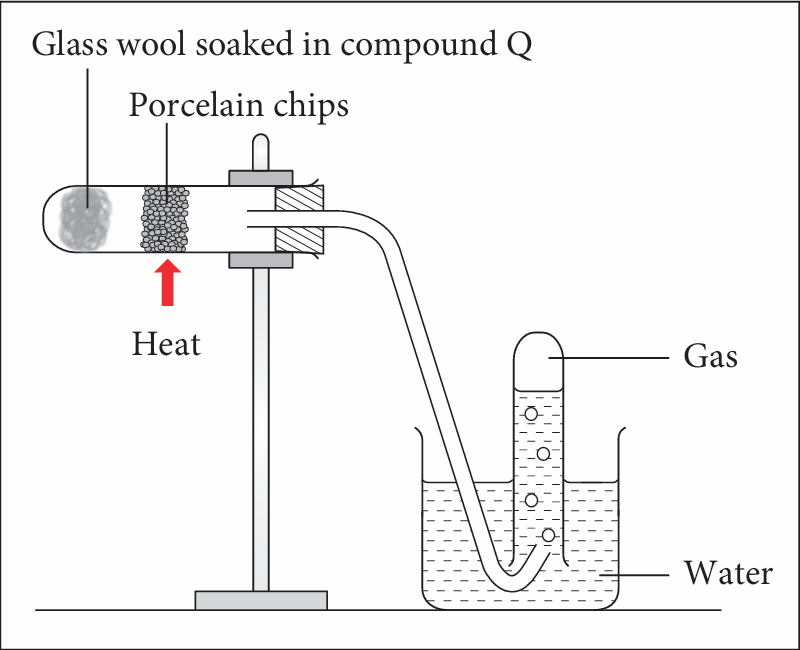
(d) Purple colour is decolourised
(e)(i) Esterification
(e)(ii) Ethyl ethanoate
