Write the complete equation for each of the following alcohol reaction.
1. Complete combustion in excess oxygen:
(a) Methanol.
(b) Propanol.
2. Oxidation by acidified potassium manganate(VII), KMnO4 oxidising agent:
(a) Butanol.
(b) Pentanol.
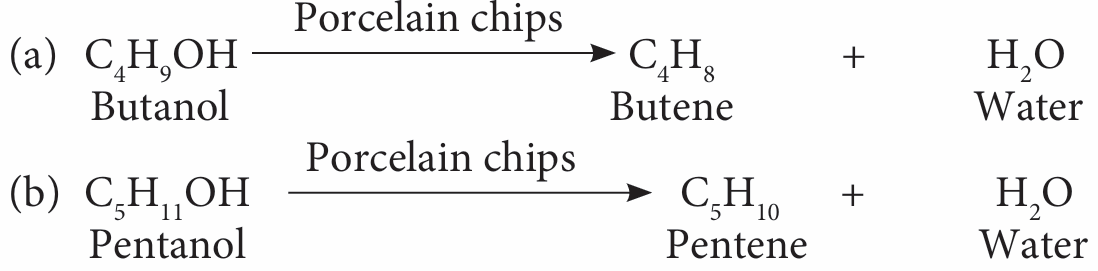
3. Dehydration by porcelain chips as the catalyst:
(a) Butanol.
(b) Pentanol.
Answer:
1. (a) 2CH3OH + 3O2 → 2CO2 + 4H2O
1. (b) 2C3H7OH + 9O2 → 6CO2 + 8H2O
2. (a)
$$
\underset{\text { Butanol }}{\mathrm{C}_4 \mathrm{H}_9 \mathrm{OH}}+2[\mathrm{O}] \rightarrow \underset{\text { Butanoic acid }}{\mathrm{C}_3 \mathrm{H}_7 \mathrm{COOH}}+\underset{\text { Water }}{\mathrm{H}_2 \mathrm{O}}
$$
2. (b)
$$
\underset{\text { Pentanol }}{\mathrm{C}_5 \mathrm{H}_{11} \mathrm{OH}}+2[\mathrm{O}] \rightarrow \underset{\text { Pentanoic acid }}{\mathrm{C}_4 \mathrm{H}_9 \mathrm{COOH}}+\underset{\text { Water }}{\mathrm{H}_2 \mathrm{O}}
$$
3.