Question 1:
The thermochemical equation for the decomposition reaction of calcium carbonate, CaCO3 is as follows:
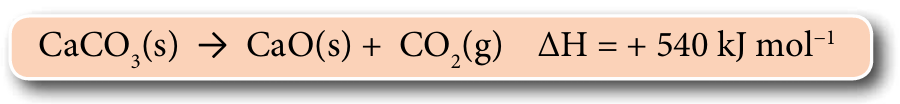
(a) Draw the energy level diagram for the reaction.
(b) Write three statements that can be obtained from the energy level diagram in (a).
Answer:
(a)
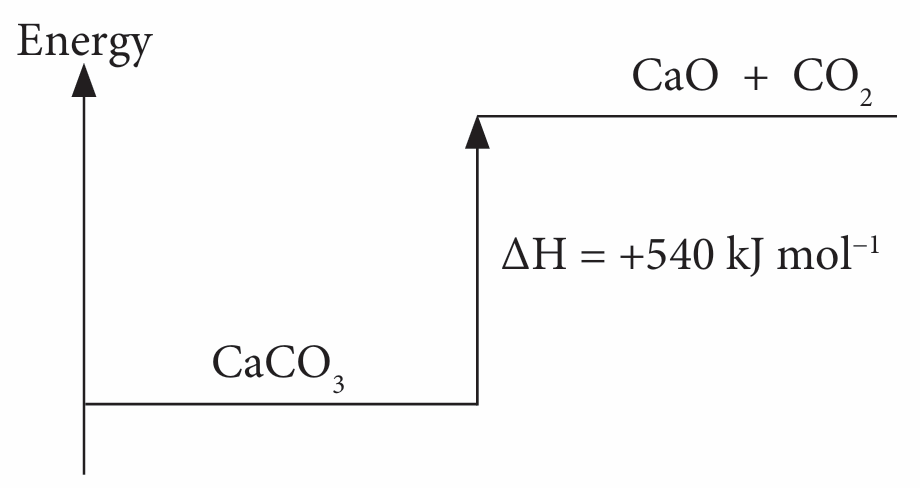
(b)
1. The decomposition of calcium carbonate, CaCO3 is an endothermic reaction.
2. When 1 mole of calcium carbonate, CaCO3 is decomposed, 540 kJ energy is absorbed from the environment.
3. The energy content of the products is higher than the energy content of the reactants.
The thermochemical equation for the decomposition reaction of calcium carbonate, CaCO3 is as follows:
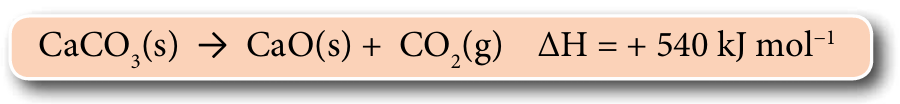
(a) Draw the energy level diagram for the reaction.
(b) Write three statements that can be obtained from the energy level diagram in (a).
Answer:
(a)

(b)
1. The decomposition of calcium carbonate, CaCO3 is an endothermic reaction.
2. When 1 mole of calcium carbonate, CaCO3 is decomposed, 540 kJ energy is absorbed from the environment.
3. The energy content of the products is higher than the energy content of the reactants.
Question 2:
The thermochemical equation below shows the formation of lead(II) sulphate, PbSO4.
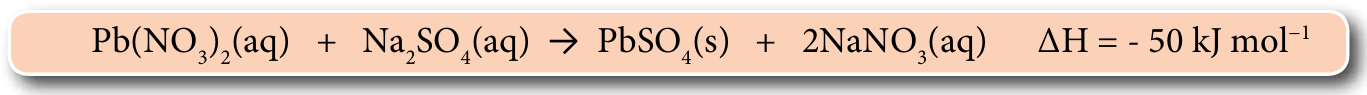
(a) State whether the reaction above is an exothermic or endothermic reaction.
(b) Determine which bond is broken and which bond is formed.
(c) Explain the energy change involved in bond breaking and bond formation.
(c) Draw the energy level diagram for the reaction.
Answer:
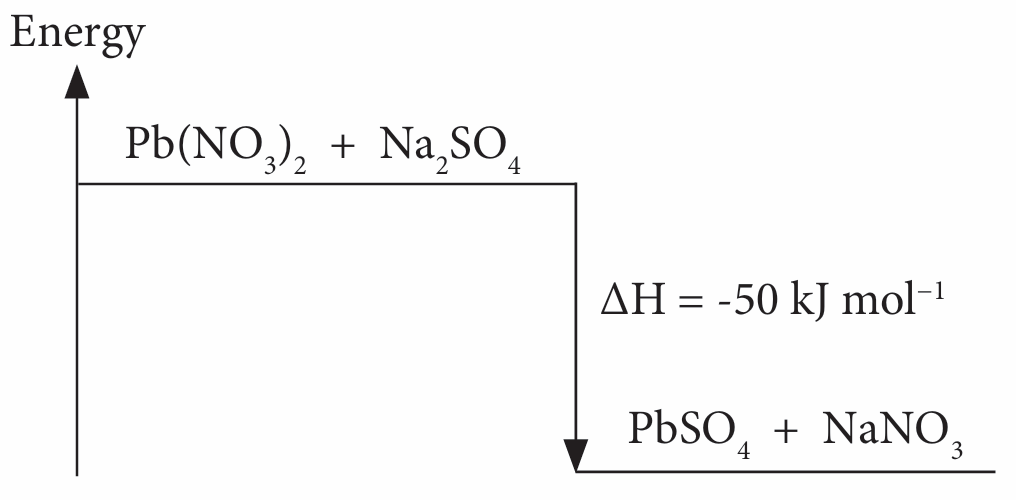
(a) Exothermic reaction
(b) Reactants (Pb(NO3)2 and Na2SO4) undergo bond breaking, while products (PbSO4 and NaNO3) undergo bond formation.
(c) Energy is absorbed during bond breaking, while heat is released during bond formation.
(d)
The thermochemical equation below shows the formation of lead(II) sulphate, PbSO4.
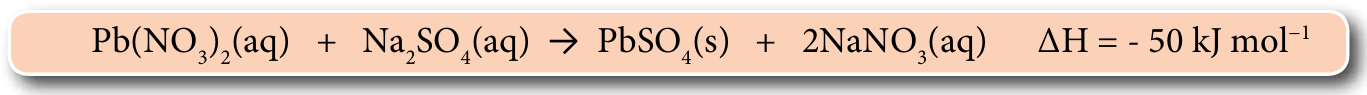
(a) State whether the reaction above is an exothermic or endothermic reaction.
(b) Determine which bond is broken and which bond is formed.
(c) Explain the energy change involved in bond breaking and bond formation.
(c) Draw the energy level diagram for the reaction.
Answer:
(a) Exothermic reaction
(b) Reactants (Pb(NO3)2 and Na2SO4) undergo bond breaking, while products (PbSO4 and NaNO3) undergo bond formation.
(c) Energy is absorbed during bond breaking, while heat is released during bond formation.
(d)

