Activity 3A:
A student carried out an experiment to determine the heat of displacement for the reaction between copper, Cu and silver nitrate, AgNO3 solution. In this experiment, excess copper powder, Cu is added into 100 cm3 of 0.5 mol dm−3 of silver nitrate, AgNO3 solution.
The heat of displacement in this experiment is -105 kJ mol−1.
[Specific heat capacity of solution, c = 4.2 J g−1 °C−1; density of solution = 1 g cm−3]
(a) Write the thermochemical equation for this reaction.
(b) Calculate the heat released in this experiment.
(c) Calculate the change in temperature.
(d) Draw the energy level diagram for this reaction.
(e) The experiment is repeated using 100 cm3 of 1.0 mol dm−3 of silver nitrate, AgNO3 solution and excess copper powder, Cu. Calculate the change in temperature in this experiment.
Explain why the change in temperature is different from the calculated value in (c).
Answer:
(a) $$ \mathrm{Cu}+2 \mathrm{AgNO}_3 \rightarrow 2 \mathrm{Ag}+\mathrm{Cu}\left(\mathrm{NO}_3\right)_2 \quad \Delta \mathrm{H}=-105 \mathrm{~kJ} \mathrm{~mol}^{-1} $$
(b)
Number of moles of silver ions
$$ =0.5 \mathrm{~mol} \mathrm{dm}^{-3} \times \frac{100}{1000} \mathrm{dm}^3=0.05 \mathrm{~mol} $$
From the thermochemical equation, 1 mole of silver displaced releases 105 kJ heat
Therefore, 0.05 moles of silver displaced releases 0.05 mol × 105 kJ mol−1 = 5.25 kJ heat
(c)
$$ \begin{aligned} & \mathrm{Q}=\mathrm{mc} \theta \text { therefore, } \theta=\frac{\mathrm{Q}}{\mathrm{mc}} \\ & \theta=\frac{5.25 \times 1000 \mathrm{~J}}{100 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1}}=12.5^{\circ} \mathrm{C} \end{aligned} $$
(d)
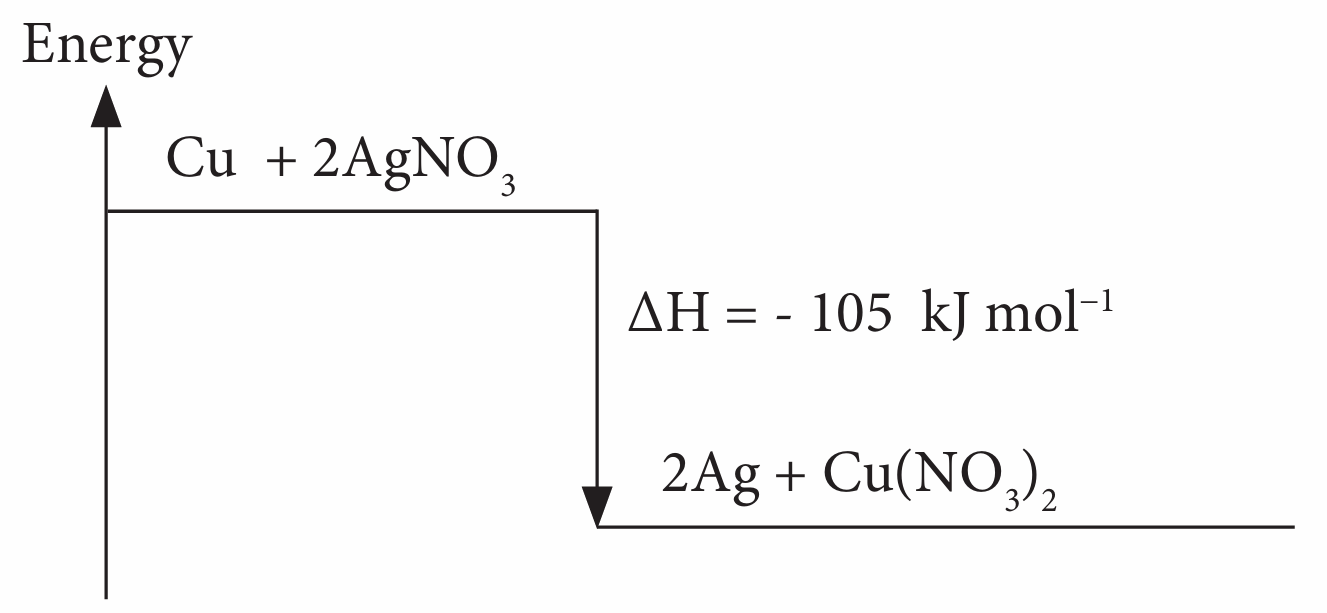
(e)
Number of moles of silver nitrate solution
$$ =1.0 \mathrm{~mol} \mathrm{dm}^{-3} \times \frac{100}{1000} \mathrm{dm}^3=0.1 \mathrm{~mol} $$
From the thermochemistry equation, displacement of 1 mole of silver releases 105 kJ of heat
Therefore, 0.1 moles of silver displaced releases 0.1 mol × 10.5 kJ mol−1 = 10.5 kJ heat
$$ \begin{aligned} & \mathrm{Q}=\mathrm{mc} \theta \text { there fore, } \theta=\frac{\mathrm{Q}}{\mathrm{mc}} \\ & \theta=\frac{10.5 \times 1000 \mathrm{~J}}{100 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1}}=25^{\circ} \mathrm{C} \end{aligned} $$
The number of moles of solution is twice, thus the change in temperature is also twice.
A student carried out an experiment to determine the heat of displacement for the reaction between copper, Cu and silver nitrate, AgNO3 solution. In this experiment, excess copper powder, Cu is added into 100 cm3 of 0.5 mol dm−3 of silver nitrate, AgNO3 solution.
The heat of displacement in this experiment is -105 kJ mol−1.
[Specific heat capacity of solution, c = 4.2 J g−1 °C−1; density of solution = 1 g cm−3]
(a) Write the thermochemical equation for this reaction.
(b) Calculate the heat released in this experiment.
(c) Calculate the change in temperature.
(d) Draw the energy level diagram for this reaction.
(e) The experiment is repeated using 100 cm3 of 1.0 mol dm−3 of silver nitrate, AgNO3 solution and excess copper powder, Cu. Calculate the change in temperature in this experiment.
Explain why the change in temperature is different from the calculated value in (c).
Answer:
(a) $$ \mathrm{Cu}+2 \mathrm{AgNO}_3 \rightarrow 2 \mathrm{Ag}+\mathrm{Cu}\left(\mathrm{NO}_3\right)_2 \quad \Delta \mathrm{H}=-105 \mathrm{~kJ} \mathrm{~mol}^{-1} $$
(b)
Number of moles of silver ions
$$ =0.5 \mathrm{~mol} \mathrm{dm}^{-3} \times \frac{100}{1000} \mathrm{dm}^3=0.05 \mathrm{~mol} $$
From the thermochemical equation, 1 mole of silver displaced releases 105 kJ heat
Therefore, 0.05 moles of silver displaced releases 0.05 mol × 105 kJ mol−1 = 5.25 kJ heat
(c)
$$ \begin{aligned} & \mathrm{Q}=\mathrm{mc} \theta \text { therefore, } \theta=\frac{\mathrm{Q}}{\mathrm{mc}} \\ & \theta=\frac{5.25 \times 1000 \mathrm{~J}}{100 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1}}=12.5^{\circ} \mathrm{C} \end{aligned} $$
(d)

(e)
Number of moles of silver nitrate solution
$$ =1.0 \mathrm{~mol} \mathrm{dm}^{-3} \times \frac{100}{1000} \mathrm{dm}^3=0.1 \mathrm{~mol} $$
From the thermochemistry equation, displacement of 1 mole of silver releases 105 kJ of heat
Therefore, 0.1 moles of silver displaced releases 0.1 mol × 10.5 kJ mol−1 = 10.5 kJ heat
$$ \begin{aligned} & \mathrm{Q}=\mathrm{mc} \theta \text { there fore, } \theta=\frac{\mathrm{Q}}{\mathrm{mc}} \\ & \theta=\frac{10.5 \times 1000 \mathrm{~J}}{100 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1}}=25^{\circ} \mathrm{C} \end{aligned} $$
The number of moles of solution is twice, thus the change in temperature is also twice.
