Combustion
- Combustion is a reaction when a substance burns completely in the excess oxygen.
- Example, combustion of methane in excess of oxygen produces carbon dioxide and water.
CH4 + 2O2 → CO2 + 2H2O - All fuel combustion are exothermic reactions.
Heat of Combustion
The Heat of Combustion of a substance is the heat energy released when 1 mole of the substance is completely burnt in excess oxygen in a standard condition
Finding Heat of Combustion
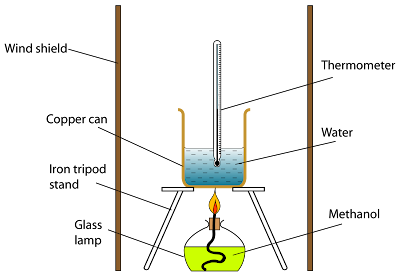
Figure Below shows a simple method for obtaining an approximate value for the heat of combustion of a fuel.
A fuel lamp is lighted and the heat energy released is used to heat a fixed volume of water. The mass of the fuel is weighed and the maximum increase of temperature is recorded.
The heat of combustion can be calculated by using the formula
It is assumed that all heat energy released from the combustion of fuel is absorbed by water.
Precaution Steps
- Use wind shield to block the air flow. This can reduce the heat loss to the surrounding.
- Do not use wire gauze, to reduce the heat loss.
- Copper container is used because copper is a good heat conductor,
- The flame must be put near the base of the container,
- The water is always stirred with the thermometer.
- The increase of water temperature is kept below 30°C to prevent over heating and energy loss to the surroundings,
- The fuel is weighed frequently to prevent condensation