Activity 3B:
When 100 cm3 of 2.0 mol dm−3 of dilute hydrochloric acid, HCl is added to 100 cm3 of 2.0 mol dm−3 of sodium hydroxide, NaOH solution, the temperature of the reaction increases from 30.0 °C to 43.5 °C.
[Specific heat capacity of solution, c = 4.2 J g−1 °C−1; density of solution = 1 g cm−3]
(a) Calculate the heat of neutralisation.
(b) Write the thermochemical equation.
(c) Construct an energy level diagram for this reaction.
(d) Predict the temperature change if the hydrochloric acid, HCl is replaced with nitric acid, HNO3 with the same volume and concentration. Explain your answer.
Answer:
(a)
$$ \begin{aligned} & \text { Mass } \mathrm{m}=(100+100) \mathrm{cm}^3 \times 1 \mathrm{~g} \mathrm{~cm}^{-3}=200 \mathrm{~g} \\ & \text { Temperature change, } \theta=(43.5-30.0)^{\circ} \mathrm{C}=13.5^{\circ} \mathrm{C} \\ & \mathrm{Q}=2000 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1} \times 13.5^{\circ} \mathrm{C}=11340 \mathrm{~J} \end{aligned} $$
Number of moles of water = Number of moles of hydrogen ions or number of moles of hydroxide ions
$$ =2.0 \mathrm{~mol} \mathrm{dm}^{-3} \times \frac{100}{1000} \mathrm{dm}^3=0.2 \mathrm{~mol} $$
Neutralisation of 0.2 moles of hydrogen ions with 0.2 moles of hydroxides ions releases 11340 J heat.
Therefore, the heat released when I mole of hydrogen ions reacts with 1 mole of hydroxide ions releases
$$ \begin{aligned} &=11340 \mathrm{~J} \times \frac{1 \mathrm{~mol}}{0.02 \mathrm{~mol}}=5670 \mathrm{~J}=56.7 \mathrm{~kJ}\\ &\text { Heat of neutralisation, } \Delta \mathrm{H}=-56.7 \mathrm{~kJ} \mathrm{~mol}^{-1} \end{aligned} $$
(b) $$ \mathrm{HCl}+\mathrm{NaOH} \rightarrow \mathrm{NaCl}+\mathrm{H}_2 \mathrm{O} \quad \Delta \mathrm{H}=-56.7 \mathrm{~kJ} \mathrm{~mol}^{-1} $$
(c)
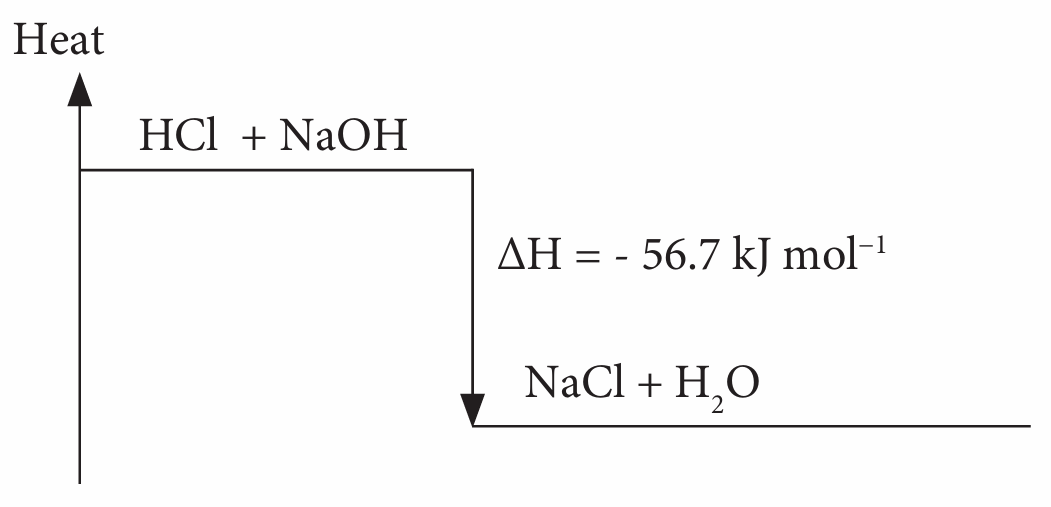
(d) Temperature change is 13.5 °C. Nitric acid is a strong acid and ionises completely in water.
When 100 cm3 of 2.0 mol dm−3 of dilute hydrochloric acid, HCl is added to 100 cm3 of 2.0 mol dm−3 of sodium hydroxide, NaOH solution, the temperature of the reaction increases from 30.0 °C to 43.5 °C.
[Specific heat capacity of solution, c = 4.2 J g−1 °C−1; density of solution = 1 g cm−3]
(a) Calculate the heat of neutralisation.
(b) Write the thermochemical equation.
(c) Construct an energy level diagram for this reaction.
(d) Predict the temperature change if the hydrochloric acid, HCl is replaced with nitric acid, HNO3 with the same volume and concentration. Explain your answer.
Answer:
(a)
$$ \begin{aligned} & \text { Mass } \mathrm{m}=(100+100) \mathrm{cm}^3 \times 1 \mathrm{~g} \mathrm{~cm}^{-3}=200 \mathrm{~g} \\ & \text { Temperature change, } \theta=(43.5-30.0)^{\circ} \mathrm{C}=13.5^{\circ} \mathrm{C} \\ & \mathrm{Q}=2000 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1} \times 13.5^{\circ} \mathrm{C}=11340 \mathrm{~J} \end{aligned} $$
Number of moles of water = Number of moles of hydrogen ions or number of moles of hydroxide ions
$$ =2.0 \mathrm{~mol} \mathrm{dm}^{-3} \times \frac{100}{1000} \mathrm{dm}^3=0.2 \mathrm{~mol} $$
Neutralisation of 0.2 moles of hydrogen ions with 0.2 moles of hydroxides ions releases 11340 J heat.
Therefore, the heat released when I mole of hydrogen ions reacts with 1 mole of hydroxide ions releases
$$ \begin{aligned} &=11340 \mathrm{~J} \times \frac{1 \mathrm{~mol}}{0.02 \mathrm{~mol}}=5670 \mathrm{~J}=56.7 \mathrm{~kJ}\\ &\text { Heat of neutralisation, } \Delta \mathrm{H}=-56.7 \mathrm{~kJ} \mathrm{~mol}^{-1} \end{aligned} $$
(b) $$ \mathrm{HCl}+\mathrm{NaOH} \rightarrow \mathrm{NaCl}+\mathrm{H}_2 \mathrm{O} \quad \Delta \mathrm{H}=-56.7 \mathrm{~kJ} \mathrm{~mol}^{-1} $$
(c)

(d) Temperature change is 13.5 °C. Nitric acid is a strong acid and ionises completely in water.
