Experiment 3A (Comparing Heat of Neutralisation):
Aim: To determine and compare heat of neutralisation between acids and alkalis of different strengths.
A. Reactions between a strong acid and a strong alkali, and a weak acid with a strong alkali.
Problem statement: Does the reaction between a strong acid and a strong alkali produce a higher heat of neutralisation when compared to the reaction between a weak acid and a strong alkali?
Hypothesis: The reaction between a strong acid and a strong alkali produces a higher heat of neutralisation than the reaction between a weak acid and a strong alkali.
Variables:
(a) Manipulated variable : Hydrochloric acid, HCl and ethanoic acid, CH3COOH
(b) Responding variable : Heat of neutralisation
(c) Fixed variable : Volume and concentration of sodium hydroxide, NaOH solution
Materials: 1.0 mol dm−3 of hydrochloric acid, HCl, 1.0 mol dm−3 of ethanoic acid, CH3COOH and 1.0 mol dm−3 of sodium hydroxide, NaOH solution.
Apparatus: Polystyrene cups with lids, thermometer and measuring cylinder.
Procedure:
1. Measure and pour 50 cm3 of 1.0 mol dm−3 of hydrochloric acid, HCl into a polystyrene cup.
2. Measure and pour 50 cm3 of 1.0 mol dm−3 of sodium hydroxide, NaOH solution into another polystyrene cup.
3. Put a thermometer into each solution and record the initial temperature of each solution.
4. Pour 1.0 mol dm−3 of hydrochloric acid, HCl quickly and carefully into the 1.0 mol dm−3 of sodium hydroxide, NaOH solution.
5. Stir the mixture using the thermometer.
6. Record the highest temperature.
7. Repeat steps 1 to 6 by replacing hydrochloric acid, HCl with ethanoic acid, CH3COOH.
B. Reactions between a strong acid and a weak alkali, and between a weak acid and a weak alkali
Problem statement: Does the reaction between a strong acid and a weak alkali produce a higher heat of neutralisation compared to the reaction between a weak acid and a weak alkali?
Hypothesis: Construct a hypothesis that involves the reactions of a strong acid, a weak acid with a weak alkali and the heat of neutralisation.
Variables: State all variables.
Materials: 1.0 mol dm−3 of hydrochloric acid, HCl, 1.0 mol dm−3 of ethanoic acid, CH3COOH and 1.0 mol dm−3 of ammonia, NH3 solution.
Apparatus: Polystyrene cups with lids, thermometer and measuring cylinder.
Procedure:
Using the materials and apparatus provided, plan and carry out an experiment to investigate the effects of strengths of acids on the heat of neutralisation with weak alkalis.
Results:
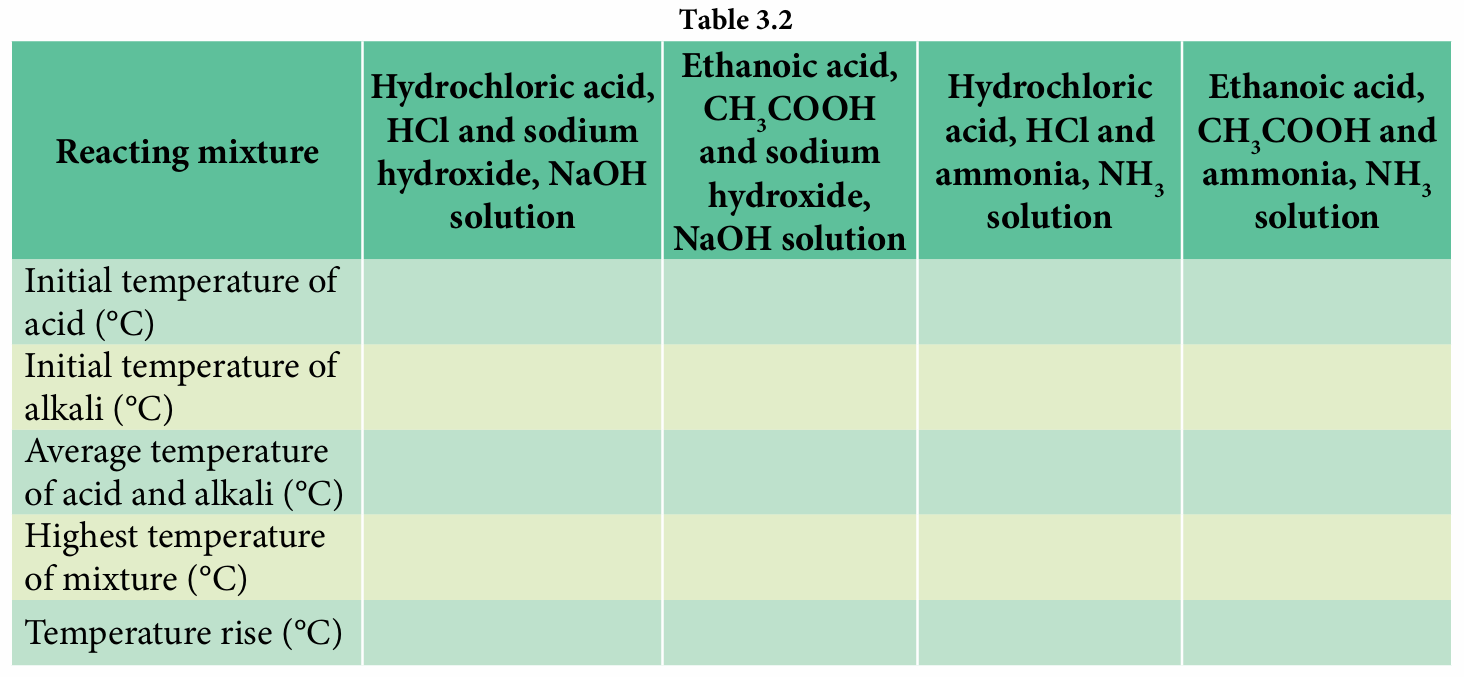
Copy and complete Table 3.2 to record the initial temperature of the acids and alkalis, average temperature of acids and alkalis, highest temperature and increase in temperature.
Discussion:
1. Write the chemical equation for each neutralisation reaction that takes place.
2. Calculate the heat of neutralisation, ∆H for each reaction.
[Given: Specific heat capacity of solution: c = 4.2 J gc °C−1; density of solution = 1 g cm−3]
3. Construct the energy level diagram for each neutralisation reaction.
4. Compare the value of the heat of neutralisation for each neutralisation reaction in this experiment.
5. Explain the reason for the difference in the value of the heat of neutralisation.
6. The theoretical value of the heat of neutralisation between a strong acid and a strong alkali is -57 kJ mol−1. Compare this value with the heat of neutralisation obtained from this experiment. Suggest a reason for this difference.
Conclusion:
Can the hypothesis be accepted? What is the conclusion of this experiment?
Answer:
Hypothesis:
The reaction between a strong acid and a weak alkali will produce a higher heat of neutralisation compared to the reaction between a weak acid and a weak alkali.
Discussions:
1.
$$ \begin{aligned} \mathrm{A}: & \mathrm{HCl}(\mathrm{aq})+\mathrm{NaOH}(\mathrm{aq}) \rightarrow \mathrm{NaCl}(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \\ & \mathrm{CH}_3 \mathrm{COOH}(\mathrm{aq})+\mathrm{NaOH}(\mathrm{aq}) \rightarrow \mathrm{CH}_3 \mathrm{COONa}(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \end{aligned} $$
$$ \begin{array}{ll} \mathrm{B}: & \mathrm{HCl}(\mathrm{aq})+\mathrm{NH}_3 \cdot \mathrm{H}_2 \mathrm{O}(\mathrm{aq}) \rightarrow \mathrm{NH}_4 \mathrm{Cl}(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \\ & \mathrm{CH}_3 \mathrm{COOH}(\mathrm{aq})+\mathrm{NH}_3 \cdot \mathrm{H}_2 \mathrm{O}(\mathrm{aq}) \rightarrow \mathrm{CH}_3 \mathrm{COONH}_4(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \end{array} $$
Note: Explanation of the neutralisation reaction using aqueous ammonia.
1. Chemical equation: HCl(aq) + NH3(aq) → NH4Cl(aq)
(NH4OH molecules do not exist)
2. Neutralisation of acids with alkalis will produce salt and water but in the case of aqueous ammonia, water in the equation is not shown.
3. Aqueous ammonia, NH3 is a weak alkali that ionises partially in water to produce hydroxide ions, OH−.
NH3(aq) + H2O(l) → NH4+(aq) + OH−(aq)
4. The neutralisation reaction between an acid and aqueous ammonia also produces water and this can be shown in the ionic equation.
5. H+ ions from the acid reacts with OH− ions to form ammonia to produce water.
Ionic equation: H+(aq) + OH−(aq) → H2O(l)
2. From the equation, 1 mole of acid reacts with 1 mole of alkali.
Steps in calculating each reaction:
(i) Calculate the number of moles of water produced.
Number of moles of H+ = Number of moles of acid
$$ =1.0 \mathrm{~mol} \mathrm{dm} \mathrm{~m}^{-3} \times \frac{50}{1000} \mathrm{dm}^3=0.05 \mathrm{~mol} \text { ion } \mathrm{H}^{+} $$
Number of moles of OH− = Number of moles of alkali
$$ =1.0 \mathrm{~mol} \mathrm{dm} \mathrm{~m}^{-3} \times \frac{50}{1000} \mathrm{dm}^3=0.05 \mathrm{~mol} \text { ion } \mathrm{OH}^{-} $$
Therefore, 0.05 moles of H+ ions react with 0.05 moles of OH− ions to produce 0.05 moles of water.
(ii) Calculate the heat change:
$$ \begin{aligned} \text { Total volume of solutions } & =\text { volume of acid }+ \text { volume of alkali } \\ & =\left(50 \mathrm{~cm}^3+50 \mathrm{~cm}^3\right)=100 \mathrm{~cm}^3 \\ \text { Mass of mixture, } \mathrm{m} & =100 \mathrm{~cm}^3 \times 1 \mathrm{~g} \mathrm{~cm}^3=100 \mathrm{~g} \end{aligned} $$
Change in temperature of the mixture, θ
Heat released in the reaction,
$$ \begin{aligned} \mathrm{Q} & =\mathrm{mc} \theta \\ & =100 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1} \times \theta=p \mathrm{~J} \end{aligned} $$
(iii) Calculate the heat of neutralisation:
0.05 moles of water formed produces p J heat
Therefore, 1 mole of water formed produces
$$ p \mathrm{~J} \times \frac{1 \mathrm{~mol}}{0.05 \mathrm{~mol}}=q \mathrm{~J}=r \mathrm{~kJ} \text { haba } $$
(iv) Write the heat of neutralisation for the reaction between an acid and an alkali as – r kJ mol−1
Note : The negative sign shows that the neutralisation reaction is exothermic.
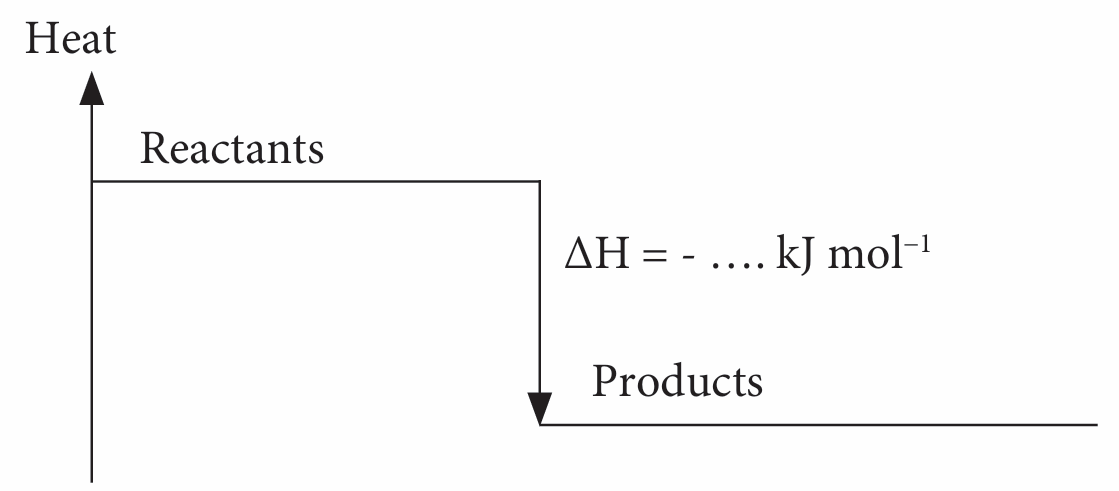
3. Construct the energy level diagram as follows:
Note: Write the chemical formulae for the reactants and the products
4. The value of the heat of neutralisation between a strong acid and and a strong alkali is higher than the heat of neutralisation between a strong acid and a weak alkali.
The value of the heat neutralisation between a strong acid and a weak alkali is higher than the heat of neutralisation between a weak acid and a weak alkali.
The value of the heat of neutralisation is the highest for the reaction between a strong acid and a strong alkali.
The value of the heat of neutralisation is the lowest for the reaction between a weak acid and a weak alkali.
5. • Hydrochloric acid is a strong acid and sodium hydroxide solution is a strong alkali. Both solutions ionise completely in water to produce a high concentration of ions.
• The neutralisation reaction involves only the combination of hydrogen ions and hydroxide ions to form water molecules.
• The heat released is not reabsorbed to ionise strong acids or strong alkalis
• Ethanoic acid is a weak acid and ionises partially in water. The concentration of hydrogen ions is low because most weak acids exist as molecules.
• During neutralisation, a small amount of heat released during the formation of 1 mole of water molecules is reabsorbed by the ethanoic acid molecules to ionise the acid molecules completely.
• Thus, when the released heat decreases, the heat of neutralisation also decreases. The same goes for aqueous ammonia solution that is a weak alkali.
6. The value of heat of neutralisation obtained in this experiment is lower. This is due to:
(a) part of the heat is released to the surroundings.
(b) the polystyrene cup absorbs heat.
Aim: To determine and compare heat of neutralisation between acids and alkalis of different strengths.
A. Reactions between a strong acid and a strong alkali, and a weak acid with a strong alkali.
Problem statement: Does the reaction between a strong acid and a strong alkali produce a higher heat of neutralisation when compared to the reaction between a weak acid and a strong alkali?
Hypothesis: The reaction between a strong acid and a strong alkali produces a higher heat of neutralisation than the reaction between a weak acid and a strong alkali.
Variables:
(a) Manipulated variable : Hydrochloric acid, HCl and ethanoic acid, CH3COOH
(b) Responding variable : Heat of neutralisation
(c) Fixed variable : Volume and concentration of sodium hydroxide, NaOH solution
Materials: 1.0 mol dm−3 of hydrochloric acid, HCl, 1.0 mol dm−3 of ethanoic acid, CH3COOH and 1.0 mol dm−3 of sodium hydroxide, NaOH solution.
Apparatus: Polystyrene cups with lids, thermometer and measuring cylinder.
Procedure:
1. Measure and pour 50 cm3 of 1.0 mol dm−3 of hydrochloric acid, HCl into a polystyrene cup.
2. Measure and pour 50 cm3 of 1.0 mol dm−3 of sodium hydroxide, NaOH solution into another polystyrene cup.
3. Put a thermometer into each solution and record the initial temperature of each solution.
4. Pour 1.0 mol dm−3 of hydrochloric acid, HCl quickly and carefully into the 1.0 mol dm−3 of sodium hydroxide, NaOH solution.
5. Stir the mixture using the thermometer.
6. Record the highest temperature.
7. Repeat steps 1 to 6 by replacing hydrochloric acid, HCl with ethanoic acid, CH3COOH.
B. Reactions between a strong acid and a weak alkali, and between a weak acid and a weak alkali
Problem statement: Does the reaction between a strong acid and a weak alkali produce a higher heat of neutralisation compared to the reaction between a weak acid and a weak alkali?
Hypothesis: Construct a hypothesis that involves the reactions of a strong acid, a weak acid with a weak alkali and the heat of neutralisation.
Variables: State all variables.
Materials: 1.0 mol dm−3 of hydrochloric acid, HCl, 1.0 mol dm−3 of ethanoic acid, CH3COOH and 1.0 mol dm−3 of ammonia, NH3 solution.
Apparatus: Polystyrene cups with lids, thermometer and measuring cylinder.
Procedure:
Using the materials and apparatus provided, plan and carry out an experiment to investigate the effects of strengths of acids on the heat of neutralisation with weak alkalis.
Results:
Copy and complete Table 3.2 to record the initial temperature of the acids and alkalis, average temperature of acids and alkalis, highest temperature and increase in temperature.

Discussion:
1. Write the chemical equation for each neutralisation reaction that takes place.
2. Calculate the heat of neutralisation, ∆H for each reaction.
[Given: Specific heat capacity of solution: c = 4.2 J gc °C−1; density of solution = 1 g cm−3]
3. Construct the energy level diagram for each neutralisation reaction.
4. Compare the value of the heat of neutralisation for each neutralisation reaction in this experiment.
5. Explain the reason for the difference in the value of the heat of neutralisation.
6. The theoretical value of the heat of neutralisation between a strong acid and a strong alkali is -57 kJ mol−1. Compare this value with the heat of neutralisation obtained from this experiment. Suggest a reason for this difference.
Conclusion:
Can the hypothesis be accepted? What is the conclusion of this experiment?
Answer:
Hypothesis:
The reaction between a strong acid and a weak alkali will produce a higher heat of neutralisation compared to the reaction between a weak acid and a weak alkali.
Discussions:
1.
$$ \begin{aligned} \mathrm{A}: & \mathrm{HCl}(\mathrm{aq})+\mathrm{NaOH}(\mathrm{aq}) \rightarrow \mathrm{NaCl}(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \\ & \mathrm{CH}_3 \mathrm{COOH}(\mathrm{aq})+\mathrm{NaOH}(\mathrm{aq}) \rightarrow \mathrm{CH}_3 \mathrm{COONa}(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \end{aligned} $$
$$ \begin{array}{ll} \mathrm{B}: & \mathrm{HCl}(\mathrm{aq})+\mathrm{NH}_3 \cdot \mathrm{H}_2 \mathrm{O}(\mathrm{aq}) \rightarrow \mathrm{NH}_4 \mathrm{Cl}(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \\ & \mathrm{CH}_3 \mathrm{COOH}(\mathrm{aq})+\mathrm{NH}_3 \cdot \mathrm{H}_2 \mathrm{O}(\mathrm{aq}) \rightarrow \mathrm{CH}_3 \mathrm{COONH}_4(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{l}) \end{array} $$
Note: Explanation of the neutralisation reaction using aqueous ammonia.
1. Chemical equation: HCl(aq) + NH3(aq) → NH4Cl(aq)
(NH4OH molecules do not exist)
2. Neutralisation of acids with alkalis will produce salt and water but in the case of aqueous ammonia, water in the equation is not shown.
3. Aqueous ammonia, NH3 is a weak alkali that ionises partially in water to produce hydroxide ions, OH−.
NH3(aq) + H2O(l) → NH4+(aq) + OH−(aq)
4. The neutralisation reaction between an acid and aqueous ammonia also produces water and this can be shown in the ionic equation.
5. H+ ions from the acid reacts with OH− ions to form ammonia to produce water.
Ionic equation: H+(aq) + OH−(aq) → H2O(l)
2. From the equation, 1 mole of acid reacts with 1 mole of alkali.
Steps in calculating each reaction:
(i) Calculate the number of moles of water produced.
Number of moles of H+ = Number of moles of acid
$$ =1.0 \mathrm{~mol} \mathrm{dm} \mathrm{~m}^{-3} \times \frac{50}{1000} \mathrm{dm}^3=0.05 \mathrm{~mol} \text { ion } \mathrm{H}^{+} $$
Number of moles of OH− = Number of moles of alkali
$$ =1.0 \mathrm{~mol} \mathrm{dm} \mathrm{~m}^{-3} \times \frac{50}{1000} \mathrm{dm}^3=0.05 \mathrm{~mol} \text { ion } \mathrm{OH}^{-} $$
Therefore, 0.05 moles of H+ ions react with 0.05 moles of OH− ions to produce 0.05 moles of water.
(ii) Calculate the heat change:
$$ \begin{aligned} \text { Total volume of solutions } & =\text { volume of acid }+ \text { volume of alkali } \\ & =\left(50 \mathrm{~cm}^3+50 \mathrm{~cm}^3\right)=100 \mathrm{~cm}^3 \\ \text { Mass of mixture, } \mathrm{m} & =100 \mathrm{~cm}^3 \times 1 \mathrm{~g} \mathrm{~cm}^3=100 \mathrm{~g} \end{aligned} $$
Change in temperature of the mixture, θ
Heat released in the reaction,
$$ \begin{aligned} \mathrm{Q} & =\mathrm{mc} \theta \\ & =100 \mathrm{~g} \times 4.2 \mathrm{~J} \mathrm{~g}^{-1}{ }^{\circ} \mathrm{C}^{-1} \times \theta=p \mathrm{~J} \end{aligned} $$
(iii) Calculate the heat of neutralisation:
0.05 moles of water formed produces p J heat
Therefore, 1 mole of water formed produces
$$ p \mathrm{~J} \times \frac{1 \mathrm{~mol}}{0.05 \mathrm{~mol}}=q \mathrm{~J}=r \mathrm{~kJ} \text { haba } $$
(iv) Write the heat of neutralisation for the reaction between an acid and an alkali as – r kJ mol−1
Note : The negative sign shows that the neutralisation reaction is exothermic.
3. Construct the energy level diagram as follows:
Note: Write the chemical formulae for the reactants and the products

4. The value of the heat of neutralisation between a strong acid and and a strong alkali is higher than the heat of neutralisation between a strong acid and a weak alkali.
The value of the heat neutralisation between a strong acid and a weak alkali is higher than the heat of neutralisation between a weak acid and a weak alkali.
The value of the heat of neutralisation is the highest for the reaction between a strong acid and a strong alkali.
The value of the heat of neutralisation is the lowest for the reaction between a weak acid and a weak alkali.
5. • Hydrochloric acid is a strong acid and sodium hydroxide solution is a strong alkali. Both solutions ionise completely in water to produce a high concentration of ions.
• The neutralisation reaction involves only the combination of hydrogen ions and hydroxide ions to form water molecules.
• The heat released is not reabsorbed to ionise strong acids or strong alkalis
• Ethanoic acid is a weak acid and ionises partially in water. The concentration of hydrogen ions is low because most weak acids exist as molecules.
• During neutralisation, a small amount of heat released during the formation of 1 mole of water molecules is reabsorbed by the ethanoic acid molecules to ionise the acid molecules completely.
• Thus, when the released heat decreases, the heat of neutralisation also decreases. The same goes for aqueous ammonia solution that is a weak alkali.
6. The value of heat of neutralisation obtained in this experiment is lower. This is due to:
(a) part of the heat is released to the surroundings.
(b) the polystyrene cup absorbs heat.
