Laboratory Activity 3C (Determining Heat of Displacement):
Aim: To determine and compare the heat of displacement of copper from copper(II) sulphate, CuSO4 solution with zinc metal, Zn and magnesium metal, Mg.
Materials: 0.5 mol dm−3 of copper(II) sulphate, CuSO4 solution, magnesium powder, Mg and zinc powder, Zn.
Apparatus: Polystyrene cups with lids, thermometer, measuring cylinder and spatula.
Procedure:
1. Based on Figure 3.9, list down the suggested apparatus and materials, plan the procedure for the experiment and consider any precautionary steps that need to be taken to calculate the heat of displacement of copper, Cu.
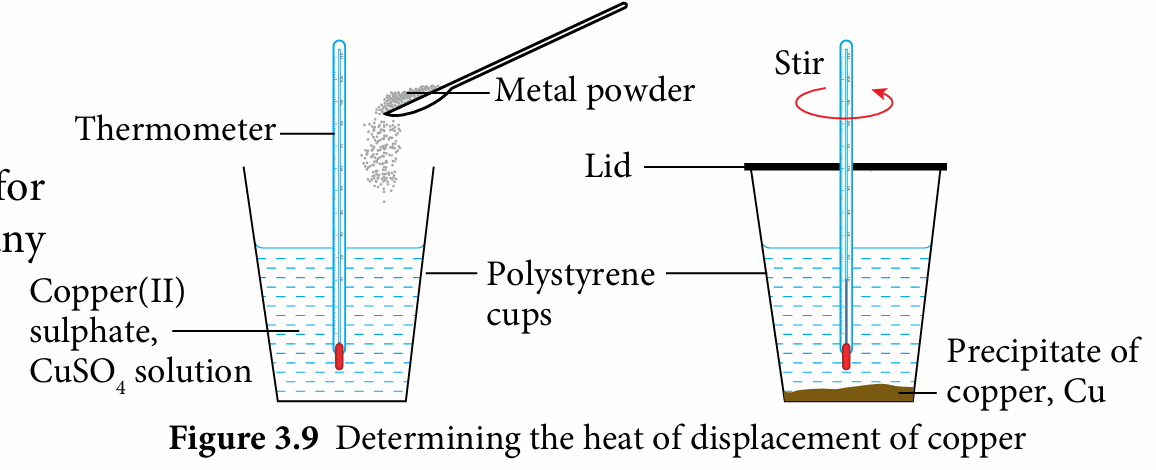
2. Discuss the planned procedures with your teacher before you proceed.
Results:
Construct a suitable table to record your readings.
Discussion:
1. Based on the experiment:
(a) write the chemical and the ionic equations for both reactions.
(b) calculate the heat of displacement, ∆H of copper, Cu by magnesium, Mg and zinc, Zn.
[Given: Specific heat capacity of solution: c = 4.2 J g−1 °C−1; density of solution = 1 g cm−3]
(c) explain why the value of heat of displacement of copper, Cu by magnesium metal, Mg and zinc metal, Zn is different.
(d) draw the energy level diagram for both reactions.
2. Why are both metals used in excess in this experiment?
3. Apart from the change in temperature, state other observations that can be found during the experiment.
4. What is the operational definition of the heat of displacement in this experiment?
5. Will the heat of displacement, ∆H of copper, Cu be the same if copper(II) sulphate, CuSO4 solution is replaced with copper(II) nitrate, Cu(NO3)2 solution? Explain your answer.
Conclusion:
Write the conclusion for this experiment.
Answer:
Procedure:
1. Measure 25 cm3 of 0.2 mol dm−3 copper(II) sulphate, CuSO4 solution and pour it into a polystyrene cup.
2. Dip a thermometer into the solution and leave aside for two minutes.
3. Record the initial temperature of the solution in a table.
4. Add one spatula of magnesium powder, Mg quickly and carefully into the polystyrene cup.
5. Close the polystyrene cup and stir the mixture using the thermometer.
6. Record the highest temperature of the mixture.
7. Repeat steps 1 to 6 by using zinc powder, Zn to replace magnesium powder, Mg.
Result:
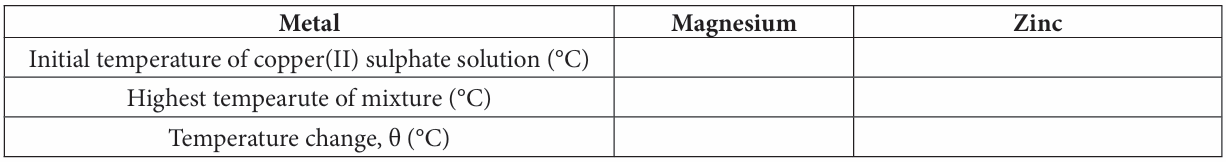
Discussions:
1. (a)
Chemical equation:
Mg(s) + CuSO4(aq) → Cu(s) + MgSO4(aq)
Zn(s) + CuSO4(aq) → Cu(s) + ZnSO4(aq)
Ionic equation:
Mg(s) + Cu2+(aq) → Cu(s) + Mg2+(aq)
Zn(s) + Cu2+(aq) → Cu(s) + Mg2+(aq)
(b) Steps in calculations:
(i) Calculate the number of moles of copper, Cu displaced from copper(II) sulphate, CuSO4 solution
Number of moles of copper(II) sulphate, CuSO4 solution
$$ =0.5 \mathrm{~mol} \mathrm{dm} \mathrm{~m}^{-3} \times \frac{25}{1000} \mathrm{dm}^3=0.0125 \mathrm{~mol} $$
From the equation, 1 mole of copper, Cu is displaced from 1 copper(II) sulphate, CuSO4 solution.
Therefore, 0.0125 moles of copper, Cu is displaced from 0.0125 moles of copper(II) sulphate, CuSO4 solution.
(ii) Calculate the heat change
Mass of solution, m = 25 cm3 × 1 g cm−3 = 25 g
Heat released in the reaction, Q = mcθ
= 25 g × 4.2 J g−1 °C−1 × θ
= x J
(iii) Calculate the heat of displacement
Displacement of 0.0125 moles of copper, Cu releases x J heat
Therefore, 1 mole of copper, Cu will release
$$ =x \mathrm{~J} \times \frac{1 \mathrm{~mol}}{0.0125 \mathrm{~mol}}=y \mathrm{~J}=z \mathrm{~kJ} \text { heat } $$
(iv) Write the heat of displacement by putting a negative sign for an exothermic reaction.
Heat of displacement of copper, Cu = – z kJ mol−1
Note: Follow the same steps to calculate the heat of displacement of copper, Cu using zinc, Zn.
(c) Magnesium is a more electropositive metal compared to zinc. Therefore the change in temperature is higher.
Thus the value of heat of precipitation is different.
(d)
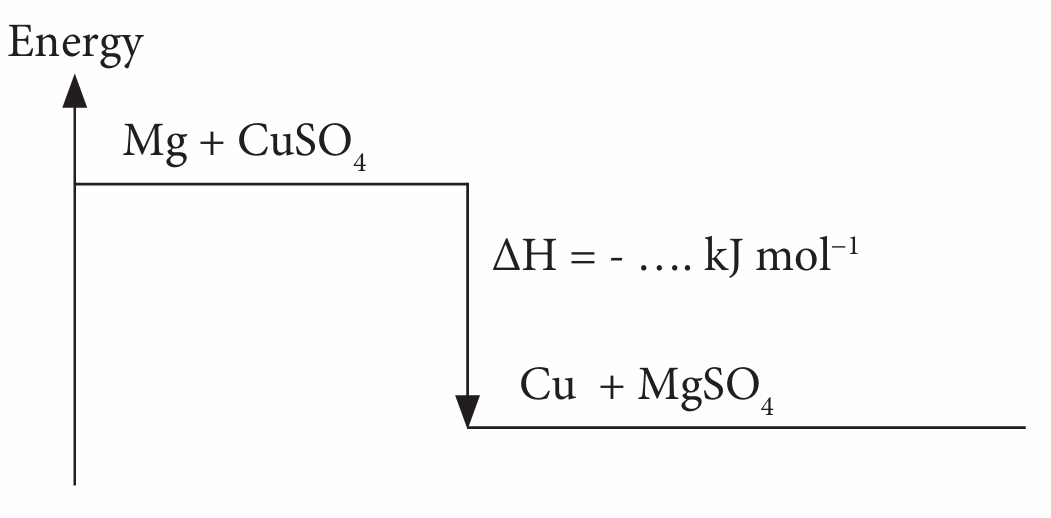
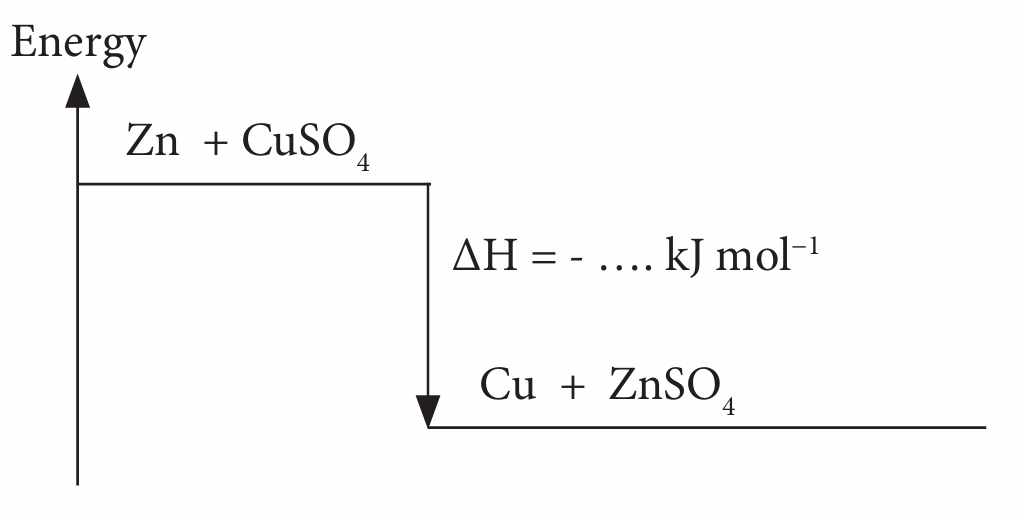
Note: Put the calculated value of ∆H
2. To ensure that all copper(II) ions are completely displaced to form copper atoms.
3. (i) Brown solid deposited.
(ii) The blue colour of the solution becomes pale blue.
4. Operational definition of heat of displacement: When magnesium metal is added into the copper(II) sulphate solution to displace 1 mole of copper, the thermometer reading increases.
5. (i) Stirring the solution slowly and continuously throughout the experiment to ensure the temperature of the mixture is uniform.
(ii) The solid metal is added quickly and carefully.
(iii) Using metal powder and not granules to ensure the reaction occurs faster.
6. Same. Heat of displacement only involves copper ions that are present in copper(II) nitrate solution.
Aim: To determine and compare the heat of displacement of copper from copper(II) sulphate, CuSO4 solution with zinc metal, Zn and magnesium metal, Mg.
Materials: 0.5 mol dm−3 of copper(II) sulphate, CuSO4 solution, magnesium powder, Mg and zinc powder, Zn.
Apparatus: Polystyrene cups with lids, thermometer, measuring cylinder and spatula.
Procedure:
1. Based on Figure 3.9, list down the suggested apparatus and materials, plan the procedure for the experiment and consider any precautionary steps that need to be taken to calculate the heat of displacement of copper, Cu.

2. Discuss the planned procedures with your teacher before you proceed.
Results:
Construct a suitable table to record your readings.
Discussion:
1. Based on the experiment:
(a) write the chemical and the ionic equations for both reactions.
(b) calculate the heat of displacement, ∆H of copper, Cu by magnesium, Mg and zinc, Zn.
[Given: Specific heat capacity of solution: c = 4.2 J g−1 °C−1; density of solution = 1 g cm−3]
(c) explain why the value of heat of displacement of copper, Cu by magnesium metal, Mg and zinc metal, Zn is different.
(d) draw the energy level diagram for both reactions.
2. Why are both metals used in excess in this experiment?
3. Apart from the change in temperature, state other observations that can be found during the experiment.
4. What is the operational definition of the heat of displacement in this experiment?
5. Will the heat of displacement, ∆H of copper, Cu be the same if copper(II) sulphate, CuSO4 solution is replaced with copper(II) nitrate, Cu(NO3)2 solution? Explain your answer.
Conclusion:
Write the conclusion for this experiment.
Answer:
Procedure:
1. Measure 25 cm3 of 0.2 mol dm−3 copper(II) sulphate, CuSO4 solution and pour it into a polystyrene cup.
2. Dip a thermometer into the solution and leave aside for two minutes.
3. Record the initial temperature of the solution in a table.
4. Add one spatula of magnesium powder, Mg quickly and carefully into the polystyrene cup.
5. Close the polystyrene cup and stir the mixture using the thermometer.
6. Record the highest temperature of the mixture.
7. Repeat steps 1 to 6 by using zinc powder, Zn to replace magnesium powder, Mg.
Result:

Discussions:
1. (a)
Chemical equation:
Mg(s) + CuSO4(aq) → Cu(s) + MgSO4(aq)
Zn(s) + CuSO4(aq) → Cu(s) + ZnSO4(aq)
Ionic equation:
Mg(s) + Cu2+(aq) → Cu(s) + Mg2+(aq)
Zn(s) + Cu2+(aq) → Cu(s) + Mg2+(aq)
(b) Steps in calculations:
(i) Calculate the number of moles of copper, Cu displaced from copper(II) sulphate, CuSO4 solution
Number of moles of copper(II) sulphate, CuSO4 solution
$$ =0.5 \mathrm{~mol} \mathrm{dm} \mathrm{~m}^{-3} \times \frac{25}{1000} \mathrm{dm}^3=0.0125 \mathrm{~mol} $$
From the equation, 1 mole of copper, Cu is displaced from 1 copper(II) sulphate, CuSO4 solution.
Therefore, 0.0125 moles of copper, Cu is displaced from 0.0125 moles of copper(II) sulphate, CuSO4 solution.
(ii) Calculate the heat change
Mass of solution, m = 25 cm3 × 1 g cm−3 = 25 g
Heat released in the reaction, Q = mcθ
= 25 g × 4.2 J g−1 °C−1 × θ
= x J
(iii) Calculate the heat of displacement
Displacement of 0.0125 moles of copper, Cu releases x J heat
Therefore, 1 mole of copper, Cu will release
$$ =x \mathrm{~J} \times \frac{1 \mathrm{~mol}}{0.0125 \mathrm{~mol}}=y \mathrm{~J}=z \mathrm{~kJ} \text { heat } $$
(iv) Write the heat of displacement by putting a negative sign for an exothermic reaction.
Heat of displacement of copper, Cu = – z kJ mol−1
Note: Follow the same steps to calculate the heat of displacement of copper, Cu using zinc, Zn.
(c) Magnesium is a more electropositive metal compared to zinc. Therefore the change in temperature is higher.
Thus the value of heat of precipitation is different.
(d)


Note: Put the calculated value of ∆H
2. To ensure that all copper(II) ions are completely displaced to form copper atoms.
3. (i) Brown solid deposited.
(ii) The blue colour of the solution becomes pale blue.
4. Operational definition of heat of displacement: When magnesium metal is added into the copper(II) sulphate solution to displace 1 mole of copper, the thermometer reading increases.
5. (i) Stirring the solution slowly and continuously throughout the experiment to ensure the temperature of the mixture is uniform.
(ii) The solid metal is added quickly and carefully.
(iii) Using metal powder and not granules to ensure the reaction occurs faster.
6. Same. Heat of displacement only involves copper ions that are present in copper(II) nitrate solution.
