Alkali Metals React with Oxygen
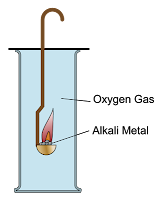
- Group 1 metals react with oxygen gas produces metal oxides. These metal oxides dissolve in water produces alkalis.
Group 1 Metals + Oxygen Gas → Metal Oxide
- Lithium, sodium and potassium form white oxide powders after reacting with oxygen.
- The white powder is the oxide of lithium, sodium and potassium.
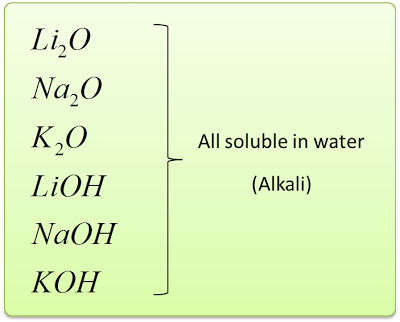
- When the white powder is dissolved in water, it produces a solution which turned red litmus paper blue. Which means, these oxides dissolve in water to form strong alkali.
- The reactivity increases down the group from lithium, sodium to potassium.
Example
Lithium + Oxygen
4Li + O2→ 2Li2O
Dissolve in water
Li2O + H2O → 2LiOH
Observation
Lithium burns with red flame and produces white powder immediately after reaction.
Sodium + Oxygen
4Na + O2→ 2Na2O
Dissolve in water
Na2O + H2O → 2NaOH
Observation
Sodium burned with bright yellow flame, forming white powder immediately after reaction.
Potassium + Oxygen
4K + O2→ 2K2O
Dissolve in water
K2O + H2O → 2KOH
Observation
Potassium burned with very bright purplish flame, forming white powder immediately after reaction.