Chemical Properties of Halogens
- Group 17 elements are very reactive non-metals.
- The atoms all have 7 valence electrons, makes them have very similar chemical properties.
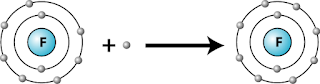
- During a chemical reaction, the atom gains one electron to form an ion with a charge of -1.
- The reactivity of group 1 decreases down the group.
- All group 17 elements are poisonous.
- Astatine is very radioactive.
Safety Precaution
- Fluorine, chlorine and bromine gases are poisonous.
- Therefore all the experiments involving these gases should be carried out in a fume chamber.
- The experiments involve fluorine are nor done in school.
- This is because fluorine is so reactive that it will react with most of the substance it comes into contact with.
- It is very difficult to conduct experiments involving fluorine.
Explaining the Reactivity Trend of the Halogens
- The reactiveness of halogens decreases down the group.
- This can be explained as below:
- When a halogen atom reacts, it gains an electron to form a singly negative charged ion.
- As we go down the group from F → Cl → Br → I, the size of the atom increases due to an extra filled electron shell.
- The valence electrons are further and further from the nucleus, the attraction force between the electrons and the nucleus become weaker and weaker.
- Therefore the ability of the atom to attract an electron to fill the outermost shell reduces., which means the reactiveness of the atom reduces.