Activity 3.13:
Aim: To determine the empirical formula of copper(II) oxide.
Materials: Water, copper(II) oxide powder, zinc granules, 1.0 mol dm–3 hydrochloric acid, wooden splinter and cotton buds
Apparatus: Boiling tube, rubber stoppers, rubber tube, 12 cm glass tube, 10 cm glass tube, spirit lamp, retort stand with clamp, wooden block, electronic balance and spatula
Procedure:
1. Weigh the mass of 12 cm glass tube using an electronic balance and record its mass.
2. Put some copper(II) oxide powder into the glass tube. Use the wooden splinter to move copper(II) oxide powder to the middle of the glass tube. Weigh the mass of the glass tube together with its contents and record the mass.
3. Fill 2/3 of the boiling tube with water.
4. Close the boiling tube with a rubber stopper that has a 12 cm glass tube. Clamp the boiling tube onto the retort stand.
5. Insert a few zinc granules into another boiling tube. Add 1.0 mol dm–3 hydrochloric acid into the boiling tube until it is 1/3 full.
6. Close the boiling tube with a rubber stopper that has a 10 cm glass tube. Clamp the boiling tube onto the other retort stand.
7. Connect the glass tube that contains copper(II) oxide powder as shown in Figure 3.13.
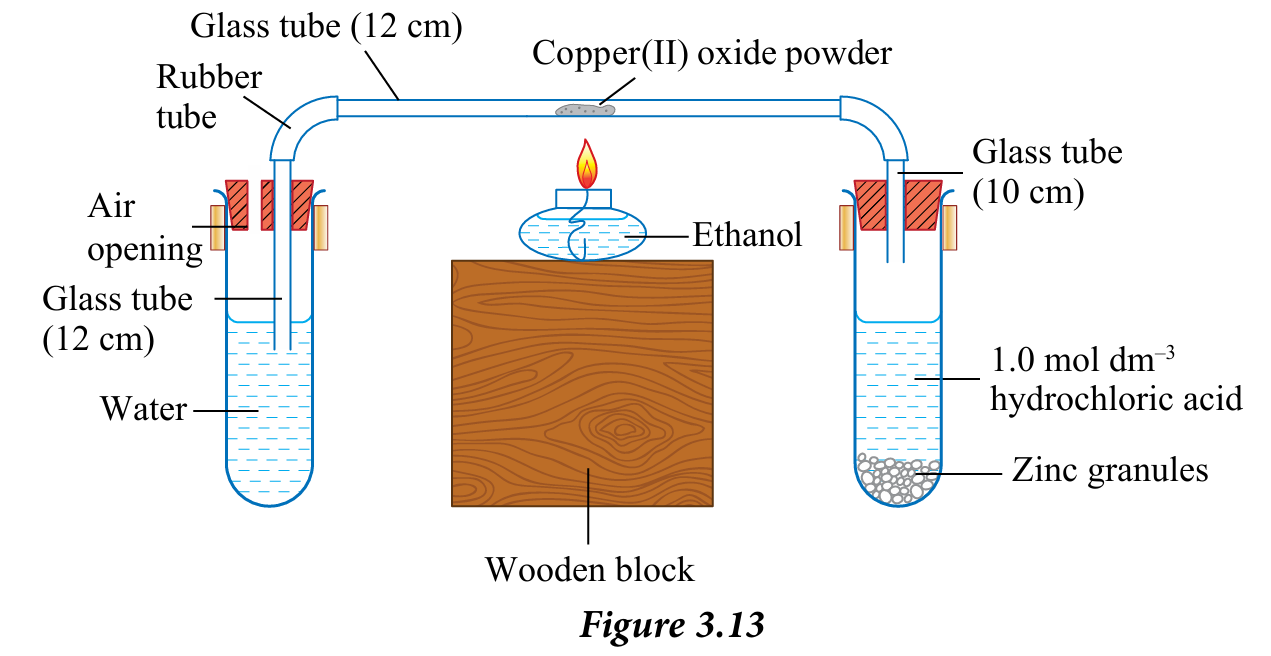
8. Let the hydrogen gas flow for 10 seconds by allowing the air bubbles to be released in the water before starting the heating process.
9. Heat copper(II) oxide using a spirit lamp with a continuous flow of hydrogen gas through the glass tube.
10. Stop the heating when the black colour of copper(II) oxide turns brown completely.
11. Keep a continuous flow of hydrogen gas until the glass tube is cooled back to room temperature.
12. Remove the glass tube that contains brown powder. Eliminate water drops at the end of the glass tube with a cotton bud.
13. Weigh the mass of the glass tube together with its contents and record its mass.
14. Repeat the heating, cooling and weighing processes from steps 9 to 13 until a constant mass reading is obtained.
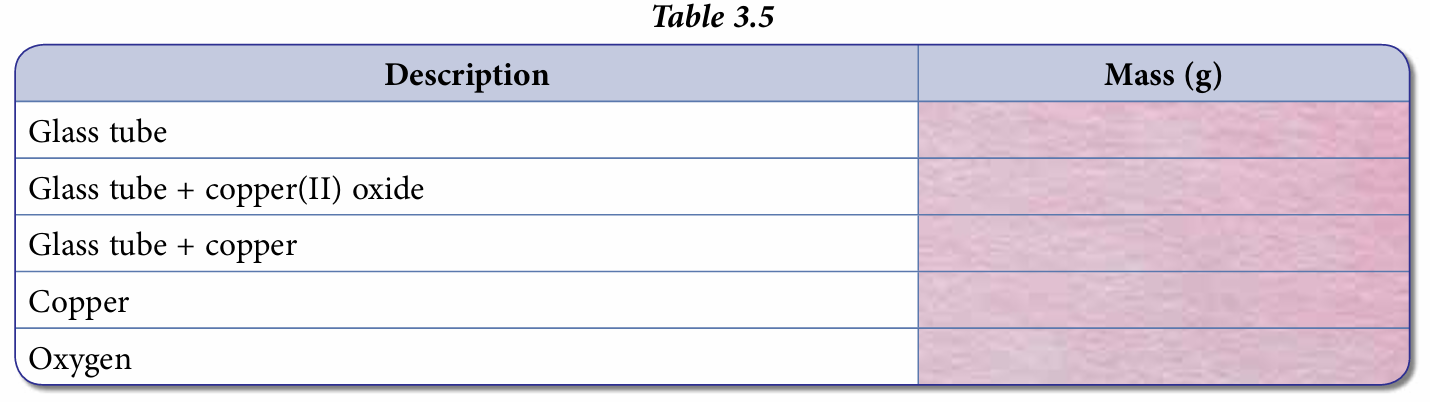
15. Record the constant mass in Table 3.5.
Results:
Interpreting data:
1. Determine the empirical formula of copper(II) oxide in this activity.
Discussion:
1. What is the purpose of using zinc granules and hydrochloric acid in this activity?
2. Why does the hydrogen gas need to flow continuously for a while before starting the heating process?
3. The hydrogen gas is allowed to flow until the product of heating is at room temperature in step 11. Why?
4. Why do the heating, cooling and weighing processes need to be repeated until a constant mass is obtained?
Answer:
Discussion:
1. To produce hydrogen gas
2. To remove air in the glass tube
3. To prevent air from entering the apparatus because oxygen in air will oxidize the hot copper back to copper(II) oxide
4. To ensure that all copper(II) oxide are completely changed to copper
Aim: To determine the empirical formula of copper(II) oxide.
Materials: Water, copper(II) oxide powder, zinc granules, 1.0 mol dm–3 hydrochloric acid, wooden splinter and cotton buds
Apparatus: Boiling tube, rubber stoppers, rubber tube, 12 cm glass tube, 10 cm glass tube, spirit lamp, retort stand with clamp, wooden block, electronic balance and spatula
Procedure:
1. Weigh the mass of 12 cm glass tube using an electronic balance and record its mass.
2. Put some copper(II) oxide powder into the glass tube. Use the wooden splinter to move copper(II) oxide powder to the middle of the glass tube. Weigh the mass of the glass tube together with its contents and record the mass.
3. Fill 2/3 of the boiling tube with water.
4. Close the boiling tube with a rubber stopper that has a 12 cm glass tube. Clamp the boiling tube onto the retort stand.
5. Insert a few zinc granules into another boiling tube. Add 1.0 mol dm–3 hydrochloric acid into the boiling tube until it is 1/3 full.
6. Close the boiling tube with a rubber stopper that has a 10 cm glass tube. Clamp the boiling tube onto the other retort stand.
7. Connect the glass tube that contains copper(II) oxide powder as shown in Figure 3.13.

8. Let the hydrogen gas flow for 10 seconds by allowing the air bubbles to be released in the water before starting the heating process.
9. Heat copper(II) oxide using a spirit lamp with a continuous flow of hydrogen gas through the glass tube.
10. Stop the heating when the black colour of copper(II) oxide turns brown completely.
11. Keep a continuous flow of hydrogen gas until the glass tube is cooled back to room temperature.
12. Remove the glass tube that contains brown powder. Eliminate water drops at the end of the glass tube with a cotton bud.
13. Weigh the mass of the glass tube together with its contents and record its mass.
14. Repeat the heating, cooling and weighing processes from steps 9 to 13 until a constant mass reading is obtained.
15. Record the constant mass in Table 3.5.
Results:

Interpreting data:
1. Determine the empirical formula of copper(II) oxide in this activity.
Discussion:
1. What is the purpose of using zinc granules and hydrochloric acid in this activity?
2. Why does the hydrogen gas need to flow continuously for a while before starting the heating process?
3. The hydrogen gas is allowed to flow until the product of heating is at room temperature in step 11. Why?
4. Why do the heating, cooling and weighing processes need to be repeated until a constant mass is obtained?
Answer:
Discussion:
1. To produce hydrogen gas
2. To remove air in the glass tube
3. To prevent air from entering the apparatus because oxygen in air will oxidize the hot copper back to copper(II) oxide
4. To ensure that all copper(II) oxide are completely changed to copper
