Question 1:
State three types of covalent bonds.
Answer:
Single bond, double bond, triple bond
State three types of covalent bonds.
Answer:
Single bond, double bond, triple bond
Question 2:
How are covalent bonds formed?
Answer:
A covalent bond is formed when non-metal atoms share electrons to achieve a stable duplet or octet electron arrangement.
How are covalent bonds formed?
Answer:
A covalent bond is formed when non-metal atoms share electrons to achieve a stable duplet or octet electron arrangement.
Question 3:
Draw the formation of covalent bonds in a water molecule, H2O.
Answer:
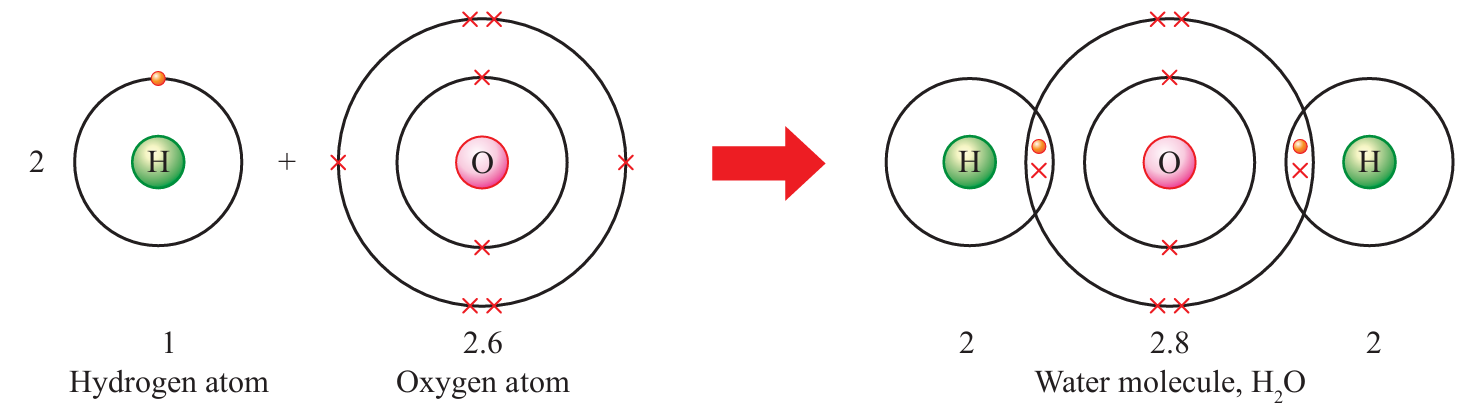
Draw the formation of covalent bonds in a water molecule, H2O.
Answer:

Question 4:
Can carbon atoms, C share electrons with four hydrogen atoms, H to form a methane molecule? Explain. [Proton number: H = 1, C = 6]
Answer:
• Yes
• The electron arrangement of carbon, C is 2.4. Carbon atom has four valence electrons. Carbon atom needs four electrons to achieve a stable octet electron arrangement, that is 2.8. Thus, carbon atom contributes four electrons to be shared.
• The electron arrangement of hydrogen, H is one. Hydrogen atom needs one electron to achieve a stable duplet electron arrangement.
• Thus, one carbon atom shares four pairs of electrons with four hydrogen atoms. Four single covalent bonds are formed and a covalent compound with formula CH4 is formed.
Can carbon atoms, C share electrons with four hydrogen atoms, H to form a methane molecule? Explain. [Proton number: H = 1, C = 6]
Answer:
• Yes
• The electron arrangement of carbon, C is 2.4. Carbon atom has four valence electrons. Carbon atom needs four electrons to achieve a stable octet electron arrangement, that is 2.8. Thus, carbon atom contributes four electrons to be shared.
• The electron arrangement of hydrogen, H is one. Hydrogen atom needs one electron to achieve a stable duplet electron arrangement.
• Thus, one carbon atom shares four pairs of electrons with four hydrogen atoms. Four single covalent bonds are formed and a covalent compound with formula CH4 is formed.
Question 5:
State one similarity and two differences between ionic bond and covalent bond.
Answer:
Only valence electrons are involved in the formation of ionic bonds and covalent bonds.
Ionic bond involves electron transfer, whereas covalent bond involves sharing of electrons.
Ionic bond involves a metal and non-metal element, whereas covalent bond involves non-metal elements only.
State one similarity and two differences between ionic bond and covalent bond.
Answer:
Only valence electrons are involved in the formation of ionic bonds and covalent bonds.
Ionic bond involves electron transfer, whereas covalent bond involves sharing of electrons.
Ionic bond involves a metal and non-metal element, whereas covalent bond involves non-metal elements only.
