Question 1:
Aluminium atom, Al has 13 protons while fluorine atom, F has 9 protons.
(a) Write the formulae of ions formed from the two atoms respectively.
(b) Write half-equations for the formation of ions in (a).
(c) Draw the electron arrangement to show the transfer of electrons in the formation of ionic bonds in aluminium fluoride compound.
Answer:
(a) Al3+ and F–
(b) Al → Al3+ + 3e–
F + e– → F–
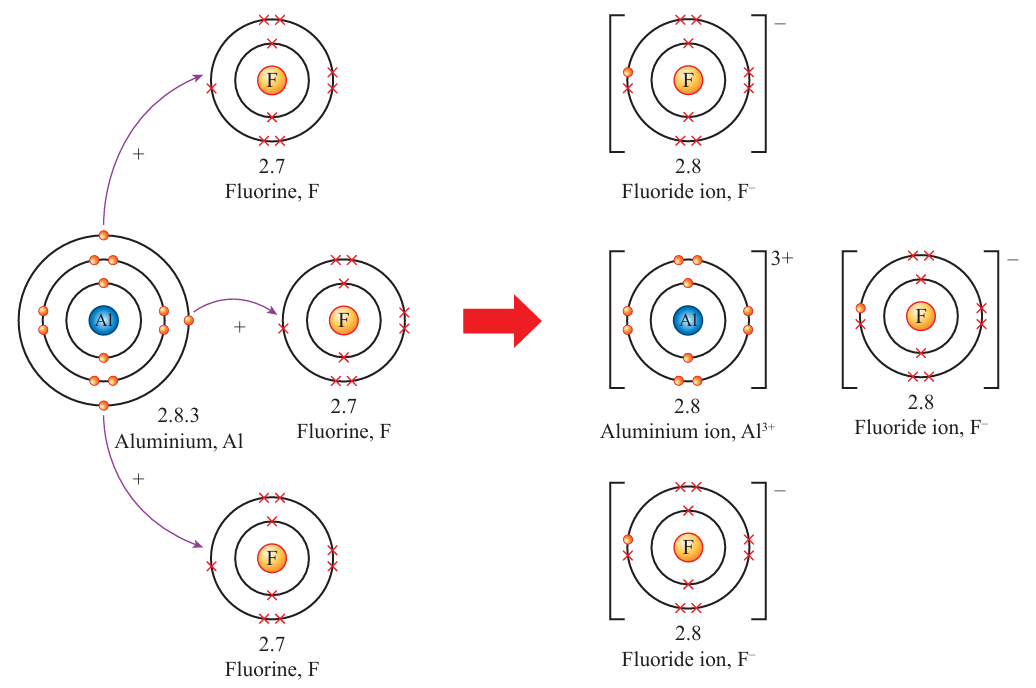
(c)
Aluminium atom, Al has 13 protons while fluorine atom, F has 9 protons.
(a) Write the formulae of ions formed from the two atoms respectively.
(b) Write half-equations for the formation of ions in (a).
(c) Draw the electron arrangement to show the transfer of electrons in the formation of ionic bonds in aluminium fluoride compound.
Answer:
(a) Al3+ and F–
(b) Al → Al3+ + 3e–
F + e– → F–
(c)

Question 2:
Muriate of Potash is a type of fertiliser that has a high content of potassium chloride compound. [Proton number: Cl = 17, K = 19]
(a) Write the chemical formula for potassium chloride.
(b) Describe the formation of ionic bonds in potassium chloride compound.
Answer:
(a) KCl
(b)
• The electron arrangement of potassium atom, K is 2.8.8.1. The potassium atom donates one electron to achieve a stable octet electron arrangement and K+ ion is formed.
• The donated electron is transferred to a chlorine atom.
• The electron arrangement of chlorine atom, Cl is 2.8.7. The chlorine atom receives one electron to form Cl– ion with a stable octet electron arrangement.
• The potassium ion, K+ and chloride ion, Cl– of opposite charges are attracted to each other by strong electrostatic attraction force.
• Potassium chloride compound, KCl is formed.
Muriate of Potash is a type of fertiliser that has a high content of potassium chloride compound. [Proton number: Cl = 17, K = 19]
(a) Write the chemical formula for potassium chloride.
(b) Describe the formation of ionic bonds in potassium chloride compound.
Answer:
(a) KCl
(b)
• The electron arrangement of potassium atom, K is 2.8.8.1. The potassium atom donates one electron to achieve a stable octet electron arrangement and K+ ion is formed.
• The donated electron is transferred to a chlorine atom.
• The electron arrangement of chlorine atom, Cl is 2.8.7. The chlorine atom receives one electron to form Cl– ion with a stable octet electron arrangement.
• The potassium ion, K+ and chloride ion, Cl– of opposite charges are attracted to each other by strong electrostatic attraction force.
• Potassium chloride compound, KCl is formed.
