Experiment 4.2:
Aim: To study the change of chemical properties of oxides of elements across Period 3.
Problem statement: How does the chemical properties of oxides of elements change across Period 3?
Materials: Sodium oxide, Na2O, magnesium oxide, MgO, aluminium oxide, Al2O3 sulphur dioxide gas, SO2, silicon(IV) oxide, SiO2, distilled water, 2.0 mol dm–3 sodium hydroxide, NaOH and 2.0 mol dm–3 nitric acid, HNO3
Apparatus: Test tube, stopper, test tube holder, 10 cm3 measuring cylinder, pH meter, Bunsen burner, glass rod and spatula
(A) Reaction of oxides of Period 3 elements with water
Hypothesis: Across Period 3, oxides of elements will change from basic to acidic.
Variables:
(a) Manipulated : Type of oxide of Period 3 elements
(b) Responding : Change in oxide property
(c) Fixed : Volume of water
Procedure:
1. Pour 10 cm3 distilled water into a test tube containing half spatula of sodium oxide, Na2O and shake.
2. Measure the pH value of the solution in the test tube using a pH meter.
3. Record your observations.
4. Repeat steps 1 to 3 using magnesium oxide, MgO, aluminium oxide, Al2O3 and sulphur dioxide, SO2.
Results:

(B) Reaction of oxides of Period 3 elements with sodium hydroxide and nitric acid
Make hypothesis and state all the variables for part B.
Procedure:
1. Fill 1/4 spatula of magnesium oxide powder, MgO into two different test tubes.
2. Add 5 cm3 of 2.0 mol dm–3 sodium hydroxide solution, NaOH into the first test tube.
3. Add 5 cm3 of 2.0 mol dm–3 nitric acid, HNO3 into the second test tube.
4. Heat both test tubes gently and stir using a glass rod as shown in Figure 4.13.
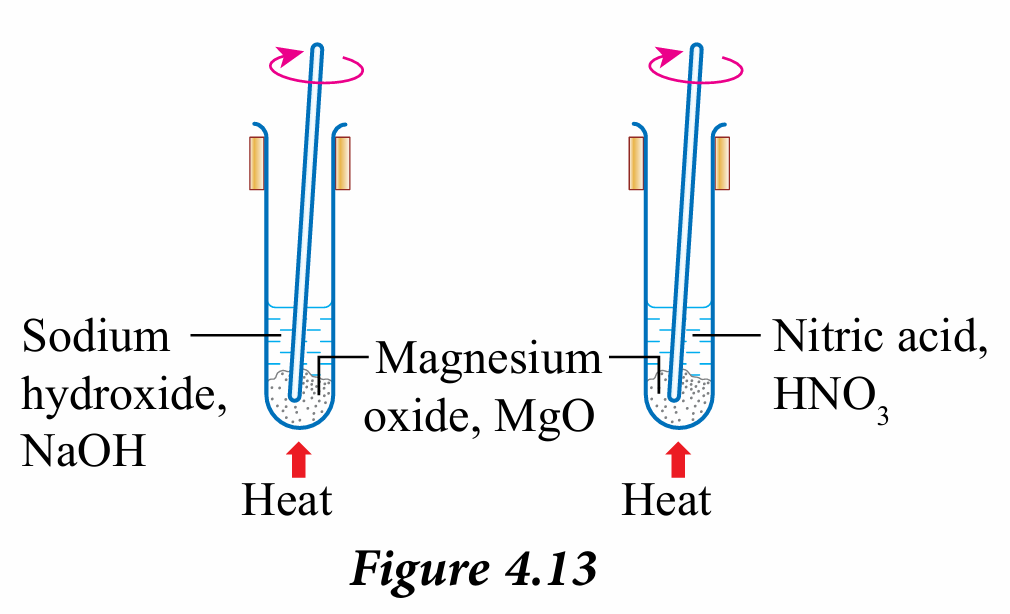
5. Observe the solubility of oxide in both solutions and record your observations.
6. Repeat steps 1 to 5 by using aluminium oxide Al2O3 and silicon(IV) oxide, SiO2.
Results:
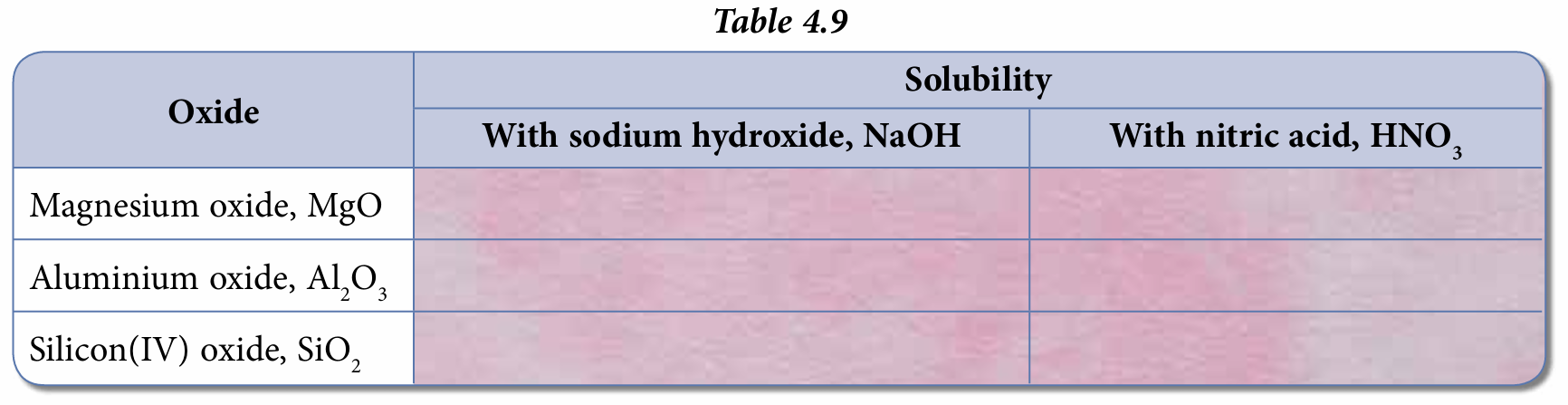
Conclusion:
Is the hypothesis acceptable? What is the conclusion of this experiment?
Discussion:
1. List the basic oxides, amphoteric oxides and acidic oxides.
2. Write the chemical equation for the reaction between basic oxide and nitric acid, HNO3.
3. Write the chemical equation for the reaction between amphoteric oxide and sodium hydroxide, NaOH.
4. List the elements that have basic oxides and acidic oxides across Period 3.
Answer:
Discussion
1.
Basic oxide: Sodium oxide, Na2O, magnesium oxide, MgO
Amphoteric oxide: Aluminium oxide, Al2O3
Acidic oxide: Silicon(IV) oxide, SiO2, sulphur dioxide, SO2
2.
MgO(s) + 2HNO3(aq) → Mg(NO3)2(aq) + H2O(l)
3.
Al2O3(s) + 2NaOH(aq) → 2NaAlO2(aq) + H2O(l)
4.
Element with basic oxide: Na and Mg
Element with acidic oxide: Si, P, S and Cl
Aim: To study the change of chemical properties of oxides of elements across Period 3.
Problem statement: How does the chemical properties of oxides of elements change across Period 3?
Materials: Sodium oxide, Na2O, magnesium oxide, MgO, aluminium oxide, Al2O3 sulphur dioxide gas, SO2, silicon(IV) oxide, SiO2, distilled water, 2.0 mol dm–3 sodium hydroxide, NaOH and 2.0 mol dm–3 nitric acid, HNO3
Apparatus: Test tube, stopper, test tube holder, 10 cm3 measuring cylinder, pH meter, Bunsen burner, glass rod and spatula
(A) Reaction of oxides of Period 3 elements with water
Hypothesis: Across Period 3, oxides of elements will change from basic to acidic.
Variables:
(a) Manipulated : Type of oxide of Period 3 elements
(b) Responding : Change in oxide property
(c) Fixed : Volume of water
Procedure:
1. Pour 10 cm3 distilled water into a test tube containing half spatula of sodium oxide, Na2O and shake.
2. Measure the pH value of the solution in the test tube using a pH meter.
3. Record your observations.
4. Repeat steps 1 to 3 using magnesium oxide, MgO, aluminium oxide, Al2O3 and sulphur dioxide, SO2.
Results:

(B) Reaction of oxides of Period 3 elements with sodium hydroxide and nitric acid
Make hypothesis and state all the variables for part B.
Procedure:
1. Fill 1/4 spatula of magnesium oxide powder, MgO into two different test tubes.
2. Add 5 cm3 of 2.0 mol dm–3 sodium hydroxide solution, NaOH into the first test tube.
3. Add 5 cm3 of 2.0 mol dm–3 nitric acid, HNO3 into the second test tube.
4. Heat both test tubes gently and stir using a glass rod as shown in Figure 4.13.

5. Observe the solubility of oxide in both solutions and record your observations.
6. Repeat steps 1 to 5 by using aluminium oxide Al2O3 and silicon(IV) oxide, SiO2.
Results:

Conclusion:
Is the hypothesis acceptable? What is the conclusion of this experiment?
Discussion:
1. List the basic oxides, amphoteric oxides and acidic oxides.
2. Write the chemical equation for the reaction between basic oxide and nitric acid, HNO3.
3. Write the chemical equation for the reaction between amphoteric oxide and sodium hydroxide, NaOH.
4. List the elements that have basic oxides and acidic oxides across Period 3.
Answer:
Discussion
1.
Basic oxide: Sodium oxide, Na2O, magnesium oxide, MgO
Amphoteric oxide: Aluminium oxide, Al2O3
Acidic oxide: Silicon(IV) oxide, SiO2, sulphur dioxide, SO2
2.
MgO(s) + 2HNO3(aq) → Mg(NO3)2(aq) + H2O(l)
3.
Al2O3(s) + 2NaOH(aq) → 2NaAlO2(aq) + H2O(l)
4.
Element with basic oxide: Na and Mg
Element with acidic oxide: Si, P, S and Cl
