Experiment 4.1:
Aim: To study the chemical properties of Group 1 elements.
Problem statement: What are the chemical properties of Group 1 elements, when they react with water, oxygen gas and chlorine gas?
Materials: Lithium, sodium, potassium, distilled water, filter paper, red litmus paper, oxygen and chlorine gas
Apparatus: Forceps, white tile, basin, knife, combustion spoon, gas jar with lid, 10 cm3 measuring cylinder and Bunsen burner
(A) Reaction of Group 1 elements with water (Demonstration by the teacher)
Hypothesis: Going down the group, the reactivity of alkali metals with water will increase.
Variables:
(a) Manipulated : Type of alkali metal
(b) Responding : Reactivity of alkali metal with water
(c) Fixed : Size of alkali metal
Procedure:
1. Cut lithium into small pieces using a knife and forceps. Dry a piece of the metal on a filter paper.
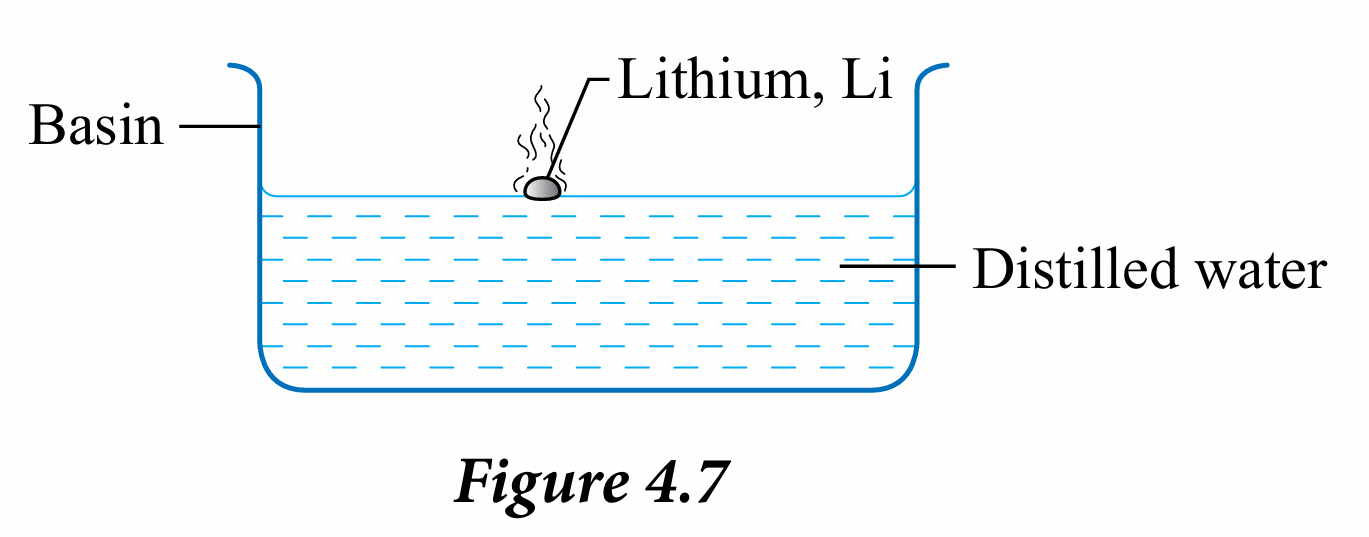
2. Put the piece of lithium slowly into a basin filled with water as shown in Figure 4.7.
3. When the reaction is complete, test the solution with a red litmus paper.
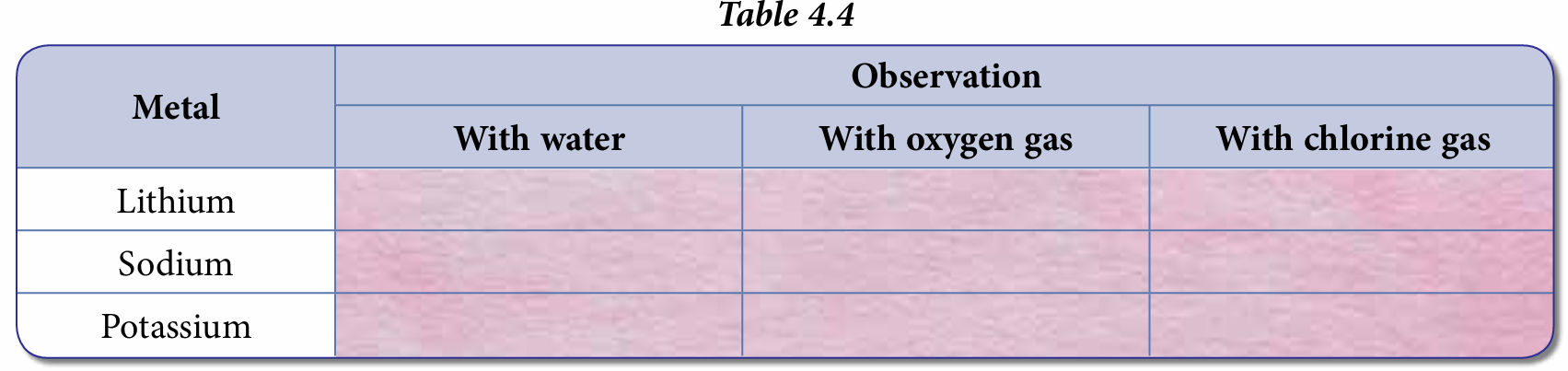
4. Record your observations in Table 4.4.
5. Repeat steps 1 to 4 with sodium and potassium.
(B) Reaction of Group 1 elements with oxygen gas
Make a hypothesis and state all the variables for part B.
Procedure:
1. Cut lithium into small pieces using a knife and forceps.
Dry a piece of the metal on a filter paper.
2. Put the piece of lithium onto a combustion spoon.
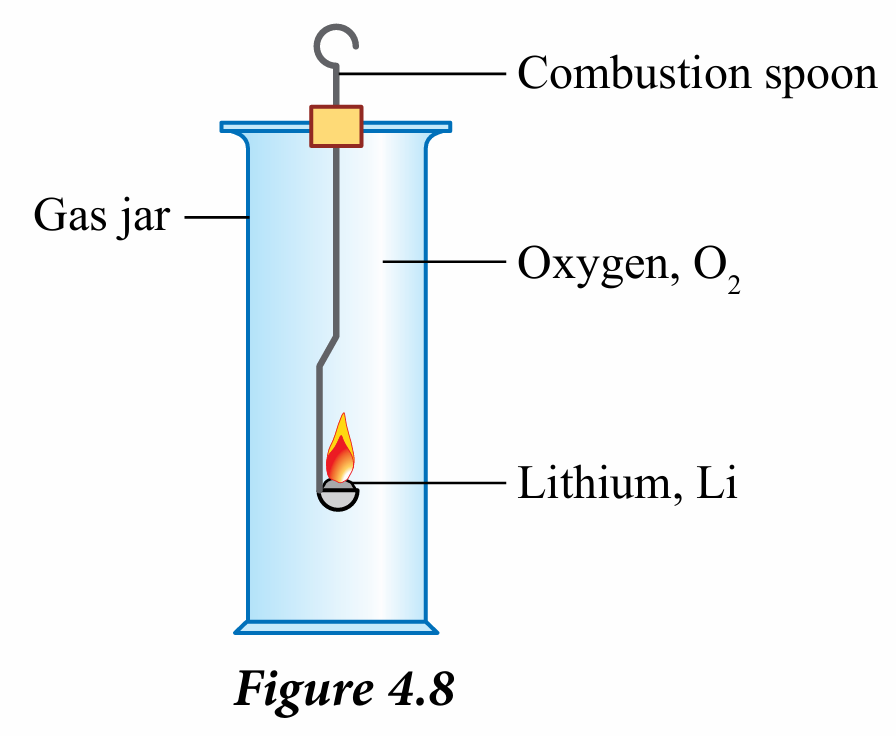
3. Heat until it starts to burn and immediately insert the spoon into a gas jar filled with oxygen gas as shown in Figure 4.8.
4. When the reaction is complete, add 10 cm3 of water into the gas jar and shake.
5. Test the solution using a red litmus paper.
6. Record your observations in Table 4.4.
7. Repeat steps 1 to 6 with sodium and potassium.
(C) Reaction of Group 1 elements with chlorine gas
Make a hypothesis and state all the variables for part C.
Procedure:
1. Cut lithium into small pieces using a knife and forceps. Dry a piece of the metal on a filter paper.
2. Put the piece of lithium onto a combustion spoon.
3. Heat until it starts to burn and immediately insert the spoon into a gas jar filled with chlorine gas as shown in Figure 4.8.
4. Record your observations in Table 4.4.
5. Repeat steps 1 to 4 with sodium and potassium.
Results:
Conclusion:
Is the hypothesis acceptable? What is the conclusion of this experiment?
Discussion:
1. Write the chemical equation for the reaction of lithium, sodium and potassium with:
(a) Water
(b) Oxygen gas
(c) Chlorine gas
2. Arrange the reactivity of alkali metals lithium, sodium and potassium with water, oxygen gas and chlorine gas in ascending order.
Answer:
Discussion
1. (a)
2Li(s) + 2H2O(l) → 2LiOH(aq) + H2(g)
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
2K(s) + 2H2O(l) → 2KOH(aq) + H2(g)
(b)
4Li(s) + O2(g) → 2Li2O(s)
4Na(s) + O2(g) → 2Na2O(s)
4K(s) + O2(g) → 2K2O(s)
(c)
2Li(s) + Cl2(g) → 2LiCl(s)
2Na(s) + Cl2(g) → 2NaCl(s)
2K(s) + Cl2(g) → 2KCl(s)
2. With water, oxygen and chlorine: Li, Na, K
Aim: To study the chemical properties of Group 1 elements.
Problem statement: What are the chemical properties of Group 1 elements, when they react with water, oxygen gas and chlorine gas?
Materials: Lithium, sodium, potassium, distilled water, filter paper, red litmus paper, oxygen and chlorine gas
Apparatus: Forceps, white tile, basin, knife, combustion spoon, gas jar with lid, 10 cm3 measuring cylinder and Bunsen burner
(A) Reaction of Group 1 elements with water (Demonstration by the teacher)
Hypothesis: Going down the group, the reactivity of alkali metals with water will increase.
Variables:
(a) Manipulated : Type of alkali metal
(b) Responding : Reactivity of alkali metal with water
(c) Fixed : Size of alkali metal
Procedure:
1. Cut lithium into small pieces using a knife and forceps. Dry a piece of the metal on a filter paper.
2. Put the piece of lithium slowly into a basin filled with water as shown in Figure 4.7.

3. When the reaction is complete, test the solution with a red litmus paper.
4. Record your observations in Table 4.4.
5. Repeat steps 1 to 4 with sodium and potassium.
(B) Reaction of Group 1 elements with oxygen gas
Make a hypothesis and state all the variables for part B.
Procedure:
1. Cut lithium into small pieces using a knife and forceps.
Dry a piece of the metal on a filter paper.
2. Put the piece of lithium onto a combustion spoon.
3. Heat until it starts to burn and immediately insert the spoon into a gas jar filled with oxygen gas as shown in Figure 4.8.

4. When the reaction is complete, add 10 cm3 of water into the gas jar and shake.
5. Test the solution using a red litmus paper.
6. Record your observations in Table 4.4.
7. Repeat steps 1 to 6 with sodium and potassium.
(C) Reaction of Group 1 elements with chlorine gas
Make a hypothesis and state all the variables for part C.
Procedure:
1. Cut lithium into small pieces using a knife and forceps. Dry a piece of the metal on a filter paper.
2. Put the piece of lithium onto a combustion spoon.
3. Heat until it starts to burn and immediately insert the spoon into a gas jar filled with chlorine gas as shown in Figure 4.8.
4. Record your observations in Table 4.4.
5. Repeat steps 1 to 4 with sodium and potassium.
Results:

Conclusion:
Is the hypothesis acceptable? What is the conclusion of this experiment?
Discussion:
1. Write the chemical equation for the reaction of lithium, sodium and potassium with:
(a) Water
(b) Oxygen gas
(c) Chlorine gas
2. Arrange the reactivity of alkali metals lithium, sodium and potassium with water, oxygen gas and chlorine gas in ascending order.
Answer:
Discussion
1. (a)
2Li(s) + 2H2O(l) → 2LiOH(aq) + H2(g)
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
2K(s) + 2H2O(l) → 2KOH(aq) + H2(g)
(b)
4Li(s) + O2(g) → 2Li2O(s)
4Na(s) + O2(g) → 2Na2O(s)
4K(s) + O2(g) → 2K2O(s)
(c)
2Li(s) + Cl2(g) → 2LiCl(s)
2Na(s) + Cl2(g) → 2NaCl(s)
2K(s) + Cl2(g) → 2KCl(s)
2. With water, oxygen and chlorine: Li, Na, K
