Question 7:
Chlorine, Cl2 reacts with sodium, Na to form a compound. Write the chemical equation for the reaction.
Answer:
2Na(s) + Cl2(g) → 2NaCl(s)
Chlorine, Cl2 reacts with sodium, Na to form a compound. Write the chemical equation for the reaction.
Answer:
2Na(s) + Cl2(g) → 2NaCl(s)
Question 8:
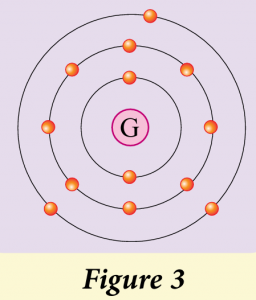
Figure 3 shows the electron arrangement for the element G.
(a) Which group does element G belong to in the Periodic Table of Elements?
(b) Which period does element G belong to in the Periodic Table of Elements?
(c) State a physical property of element G.
Answer:
(a) Group 13
(b) Period 3
(c) G is a metal

Figure 3 shows the electron arrangement for the element G.
(a) Which group does element G belong to in the Periodic Table of Elements?
(b) Which period does element G belong to in the Periodic Table of Elements?
(c) State a physical property of element G.
Answer:
(a) Group 13
(b) Period 3
(c) G is a metal
Question 9:
(a) State which element is a metal, metalloid or non-metal from the list of elements given above.
(b) Explain the change in atomic radius across the Period from left to right.
(c) Which element is a noble gas?
(d) Write the chemical equation when a metal reacts with water.
Answer:
(a)
Metal: Na, Mg, Al
Metalloid: Si
Non-metal: P, S, Cl, Ar
(b) The atomic radius decreases from Na to Cl. This is because as the number of electrons increases, the number of protons also increases. Hence the nuclear charge increases and its attraction towards the electrons also increases causing the electronegativity to increase. Hence, the size of the atom decreases as the atomic radius to decreases.
(c) Argon
(d) 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)

(a) State which element is a metal, metalloid or non-metal from the list of elements given above.
(b) Explain the change in atomic radius across the Period from left to right.
(c) Which element is a noble gas?
(d) Write the chemical equation when a metal reacts with water.
Answer:
(a)
Metal: Na, Mg, Al
Metalloid: Si
Non-metal: P, S, Cl, Ar
(b) The atomic radius decreases from Na to Cl. This is because as the number of electrons increases, the number of protons also increases. Hence the nuclear charge increases and its attraction towards the electrons also increases causing the electronegativity to increase. Hence, the size of the atom decreases as the atomic radius to decreases.
(c) Argon
(d) 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
Question 10:
Give the colour of the following transition element ions:
(a) Iron(II) ion
(b) Iron(III) ion
Answer:
(a) Green
(b) Brown
Give the colour of the following transition element ions:
(a) Iron(II) ion
(b) Iron(III) ion
Answer:
(a) Green
(b) Brown
