Question:
Refer to the Data Table of Elements on page 276.
[Avogadro constant, NA : 6.02 × 1023 mol–1; Molar volume = 22.4 dm3 mol–1 at STP or 24 dm3 mol–1 at room conditions]
1. What is meant by molar mass and molar volume?
2. What is the relationship between Avogadro constant, number of particles and number of moles?
3. Relative atomic mass of nitrogen is 14
State the meaning of the above statement based on the carbon-12 scale.
4. Vitamin C or ascorbic acid is an important antioxidant required for our health. Vitamin C has the molecular formula C6H8O6.
(a) What is the empirical formula of vitamin C?
(b) What is the relative molecular mass of vitamin C?
5. Antacid functions to relieve gastric problems. Figure 1 shows the label on a bottle of antacid.
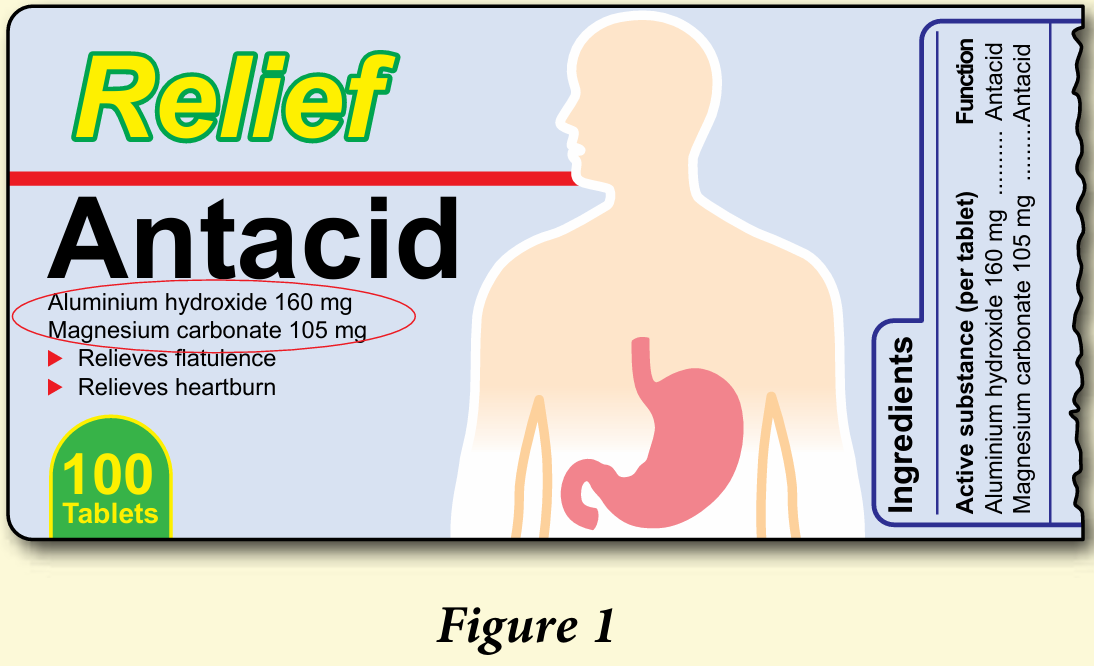
Give the chemical formulae of the two active ingredients in the antacid.
Answer:
1. Molar mass is the mass of one mole of substance in gram
Molar volume is the volume of one mole of gas
2. Number of particles in a substance = Avogadro’s constant, NA × Number of moles of substance
$$ \left(\text { or number of moles of substance }=\frac{\text { Number of particles in a substance }}{\text { Avogadro’s constant, } N_{\mathrm{A}}}\right) $$
3. Mass of one nitrogen atom is 14 times larger than 1/12 mass of one carbon-12 atom
4. (a) C3H4O3
(b) 176
5. Al(OH)3 and MgCO3
Refer to the Data Table of Elements on page 276.
[Avogadro constant, NA : 6.02 × 1023 mol–1; Molar volume = 22.4 dm3 mol–1 at STP or 24 dm3 mol–1 at room conditions]
1. What is meant by molar mass and molar volume?
2. What is the relationship between Avogadro constant, number of particles and number of moles?
3. Relative atomic mass of nitrogen is 14
State the meaning of the above statement based on the carbon-12 scale.
4. Vitamin C or ascorbic acid is an important antioxidant required for our health. Vitamin C has the molecular formula C6H8O6.
(a) What is the empirical formula of vitamin C?
(b) What is the relative molecular mass of vitamin C?
5. Antacid functions to relieve gastric problems. Figure 1 shows the label on a bottle of antacid.

Give the chemical formulae of the two active ingredients in the antacid.
Answer:
1. Molar mass is the mass of one mole of substance in gram
Molar volume is the volume of one mole of gas
2. Number of particles in a substance = Avogadro’s constant, NA × Number of moles of substance
$$ \left(\text { or number of moles of substance }=\frac{\text { Number of particles in a substance }}{\text { Avogadro’s constant, } N_{\mathrm{A}}}\right) $$
3. Mass of one nitrogen atom is 14 times larger than 1/12 mass of one carbon-12 atom
4. (a) C3H4O3
(b) 176
5. Al(OH)3 and MgCO3
