Activity 3.2:
Studying the concepts of relative atomic mass and relative molecular mass by analogy
Materials: 36 washers, one 5 cm bolt, five nuts, one flat magnet and strings
Apparatus: Two-pan balance

(A) Relative atomic mass based on the carbon-12 scale
Procedure:
1. You are given three models of carbon-12 atom. Calculate the number of washers required to form each model of carbon-12 atom.
2. Separate the washers in each model and use them for the following steps.
3. Place an atom of element A on a two-pan balance as shown in Figure 3.4.
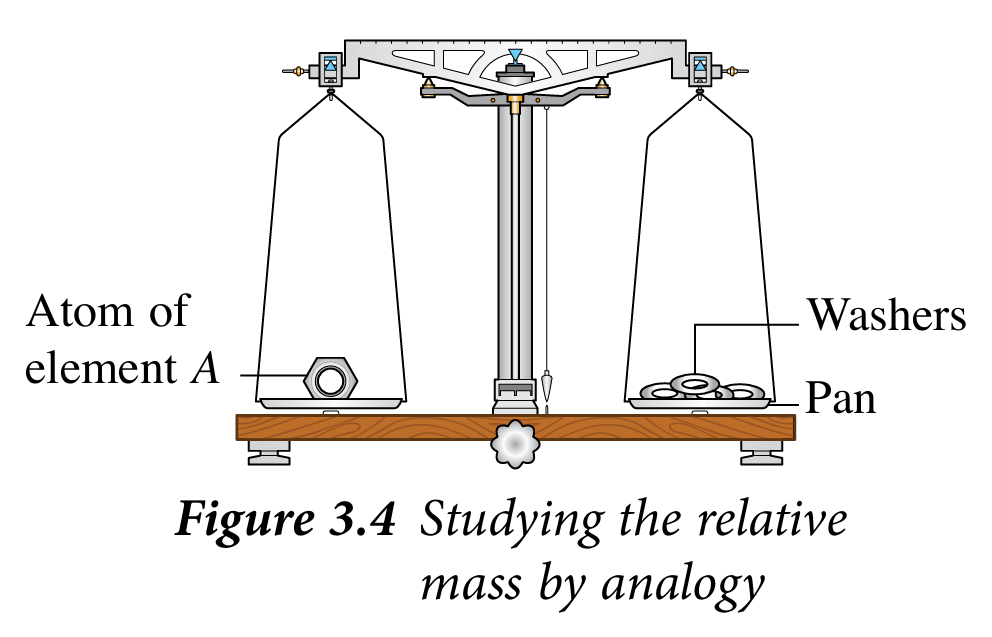 4. Place the washers on the other pan one by one until they are balanced.
4. Place the washers on the other pan one by one until they are balanced.
5. Count and record the number of washers used in Table 3.1.
6. Repeat steps 3 to 5 using atom of element B and atom of element C.
7. Calculate the relative mass of each washer in the model by assuming that each atom of carbon-12 is given the accurate mass of 12 units. Then, deduce the relative atomic masses of elements A, B and C.
Results:

Discussion:
1. How many washers form a model of carbon-12 atom?
2. What is represented by 1/12 of the mass of carbon-12 atom in this activity?
3. Define the relative atomic mass of an element based on the carbon-12 scale.
(B) Relative molecular mass based on the carbon-12 scale
Procedure:
1. Prepare models of molecules X, Y and Z as in Photograph 3.3.
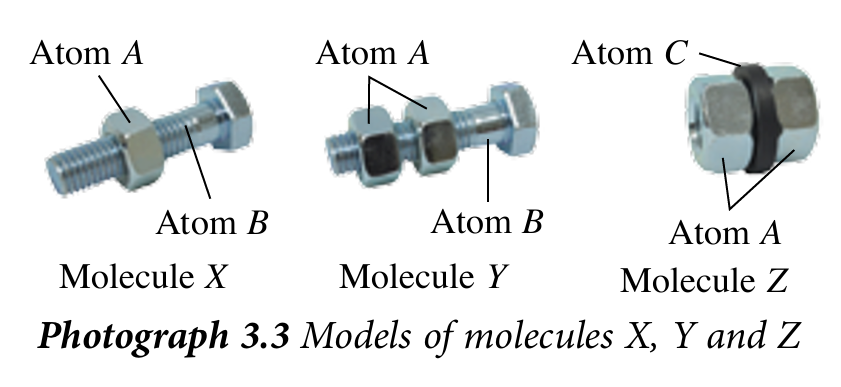
2. Place molecule X on one of the pans of the balance.
3. Place washers on the other pan one by one until they are balanced.
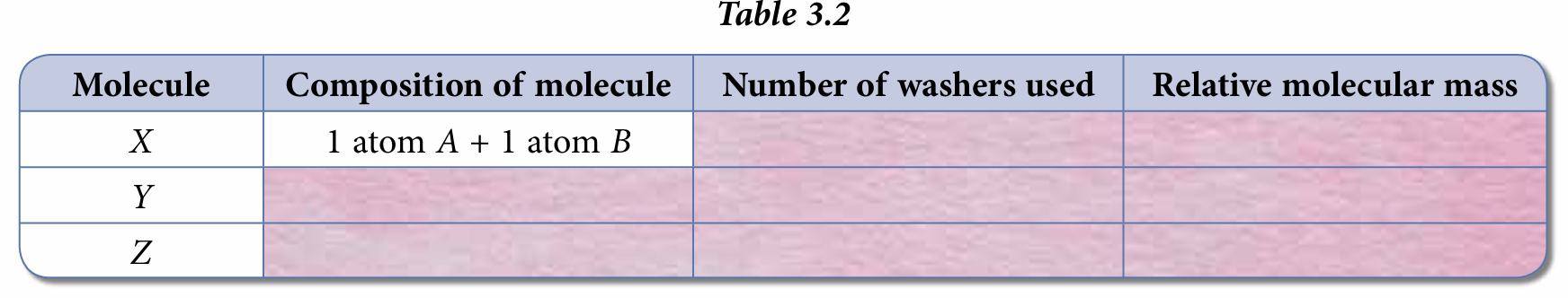
4. Count and record the number of washers used in Table 3.2.
5. Repeat steps 2 to 4 using molecule Y and Z.
6. Deduce the relative molecular masses of X, Y and Z.
Results:
Discussion:
1. Based on Activity B, give the definition of relative molecular mass based on the carbon-12 scale.
2. Calculate the relative atomic masses of all the elements that form molecules X, Y and Z.
3. Compare the answers from question 2 with the relative molecular masses you obtained in Activity B. What inference can you make about the relationship between the relative molecular mass and the relative atomic mass?
4. Molecule W is formed from one atom of element A, one atom of element B and one atom of element C. Predict the relative molecular mass of W.
Answer:
A. Discussion:
1. 12
2. Washer
3. Average mass of one atom of an element compared to 1/12 mass of one atom of carbon-12
B. Discussion:
1. Average mass of one molecule compared to 1/12 mass of one atom of carbon-12
2. (Student’s suitable answer)
3. Relative molecular mass is the sum of all relative atomic masses of all atoms that form a molecule
4. (Student’s suitable answer using the following relationship:
RAM of molecule W = RAM of A + RAM of B + RAM of C)
Studying the concepts of relative atomic mass and relative molecular mass by analogy
Materials: 36 washers, one 5 cm bolt, five nuts, one flat magnet and strings
Apparatus: Two-pan balance

(A) Relative atomic mass based on the carbon-12 scale
Procedure:
1. You are given three models of carbon-12 atom. Calculate the number of washers required to form each model of carbon-12 atom.
2. Separate the washers in each model and use them for the following steps.
3. Place an atom of element A on a two-pan balance as shown in Figure 3.4.
 4. Place the washers on the other pan one by one until they are balanced.
4. Place the washers on the other pan one by one until they are balanced. 5. Count and record the number of washers used in Table 3.1.
6. Repeat steps 3 to 5 using atom of element B and atom of element C.
7. Calculate the relative mass of each washer in the model by assuming that each atom of carbon-12 is given the accurate mass of 12 units. Then, deduce the relative atomic masses of elements A, B and C.
Results:

Discussion:
1. How many washers form a model of carbon-12 atom?
2. What is represented by 1/12 of the mass of carbon-12 atom in this activity?
3. Define the relative atomic mass of an element based on the carbon-12 scale.
(B) Relative molecular mass based on the carbon-12 scale
Procedure:
1. Prepare models of molecules X, Y and Z as in Photograph 3.3.

2. Place molecule X on one of the pans of the balance.
3. Place washers on the other pan one by one until they are balanced.
4. Count and record the number of washers used in Table 3.2.
5. Repeat steps 2 to 4 using molecule Y and Z.
6. Deduce the relative molecular masses of X, Y and Z.
Results:

Discussion:
1. Based on Activity B, give the definition of relative molecular mass based on the carbon-12 scale.
2. Calculate the relative atomic masses of all the elements that form molecules X, Y and Z.
3. Compare the answers from question 2 with the relative molecular masses you obtained in Activity B. What inference can you make about the relationship between the relative molecular mass and the relative atomic mass?
4. Molecule W is formed from one atom of element A, one atom of element B and one atom of element C. Predict the relative molecular mass of W.
Answer:
A. Discussion:
1. 12
2. Washer
3. Average mass of one atom of an element compared to 1/12 mass of one atom of carbon-12
B. Discussion:
1. Average mass of one molecule compared to 1/12 mass of one atom of carbon-12
2. (Student’s suitable answer)
3. Relative molecular mass is the sum of all relative atomic masses of all atoms that form a molecule
4. (Student’s suitable answer using the following relationship:
RAM of molecule W = RAM of A + RAM of B + RAM of C)
