Question:
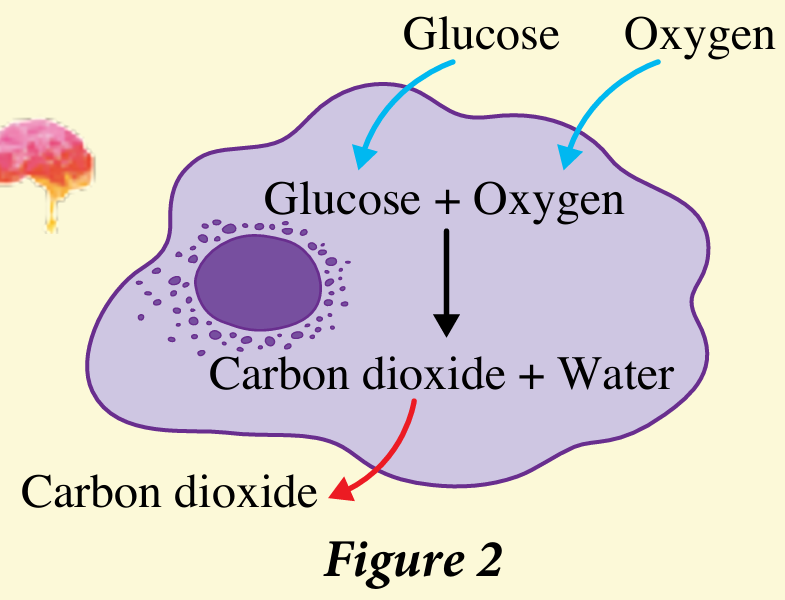
6. Figure 2 shows the aerobic respiration in our body cells to produce energy from glucose, C6H12O6. Write a balanced chemical equation for the process of aerobic respiration.
7. Iron(II) sulphate heptahydrate, FeSO4.7H2O is often used to treat anaemic patients suffering from the lack of iron mineral.
(a) What is the molar mass of iron(II) sulphate heptahydrate?
(b) Calculate the percentage of iron in iron(II) sulphate heptahydrate.
8. Figure 3 shows the weighing steps taken in the determination of the empirical formula of the oxide of metal Y.
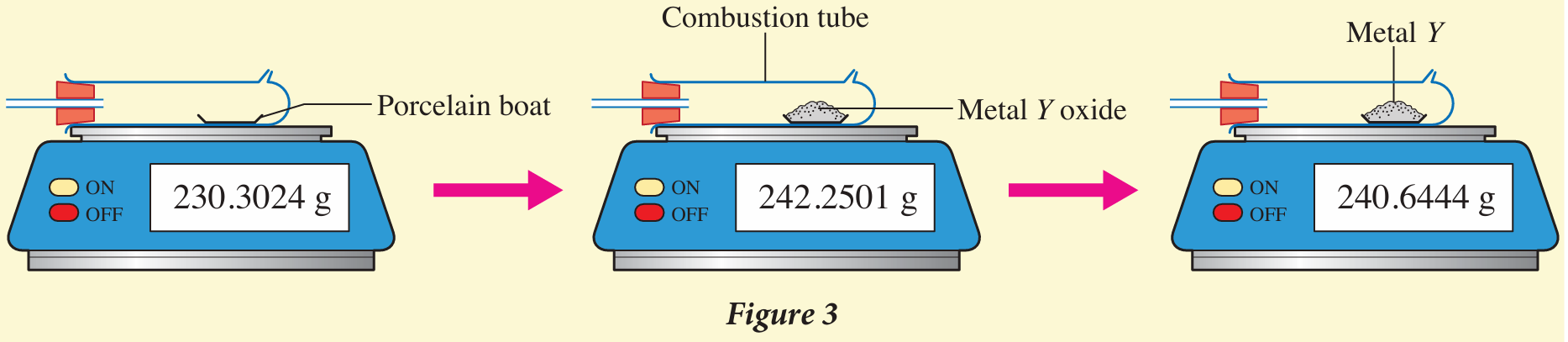
Determine the empirical formula of metal Y oxide.
[Relative atomic mass: O = 16, Y = 207]
9. P, Q and R are three samples of chemical substances.

Arrange the three samples in ascending order of mass.
10. In your opinion, between the empirical formula and the molecular formula, which formula is more suitable to be used when writing chemical equations? Give your reasons.
Answer:
6. C6H12O6(s) + 6O2(g) → 6CO2(g) + 6H2O(l)
7. (a) 278 g mol–1
(b) 20.14%
8. YO2
9. Q, R, P
10. Molecular formula is more suitable to be used in an equation because a molecular formula shows the actual number of each type of atom in a molecular compound. Hence, a stoichiometric equation that uses molecular formula can give the actual mole ratio of reactants involved. Hence, it can be used to solve various numerical reaction problems.
6. Figure 2 shows the aerobic respiration in our body cells to produce energy from glucose, C6H12O6. Write a balanced chemical equation for the process of aerobic respiration.

7. Iron(II) sulphate heptahydrate, FeSO4.7H2O is often used to treat anaemic patients suffering from the lack of iron mineral.
(a) What is the molar mass of iron(II) sulphate heptahydrate?
(b) Calculate the percentage of iron in iron(II) sulphate heptahydrate.
8. Figure 3 shows the weighing steps taken in the determination of the empirical formula of the oxide of metal Y.

Determine the empirical formula of metal Y oxide.
[Relative atomic mass: O = 16, Y = 207]
9. P, Q and R are three samples of chemical substances.

Arrange the three samples in ascending order of mass.
10. In your opinion, between the empirical formula and the molecular formula, which formula is more suitable to be used when writing chemical equations? Give your reasons.
Answer:
6. C6H12O6(s) + 6O2(g) → 6CO2(g) + 6H2O(l)
7. (a) 278 g mol–1
(b) 20.14%
8. YO2
9. Q, R, P
10. Molecular formula is more suitable to be used in an equation because a molecular formula shows the actual number of each type of atom in a molecular compound. Hence, a stoichiometric equation that uses molecular formula can give the actual mole ratio of reactants involved. Hence, it can be used to solve various numerical reaction problems.
